Le score d'ApgarBulletin WHEC pratique et de directives cliniques de gestion pour les fournisseurs de soins de santé. Subvention à l'éducation fournie par la santé des femmes et de l'Education Center (WHEC). The Apgar score, originally designed as a quick way to assess the status of the newborn infant, is used in virtually every delivery room. This universal application is a testimony to its simplicity and practical value. Over the years, however, it came to be assigned a much broader role in the prediction of long-term outcome of infants, neurologic outcome. Although there was little scientific evidence to support the use of the Apgar score in this way, its validity was widely assumed, even within the medical community. Evidence of this trend was provided in 1983 study that also described the associated misconception. The Apgar score is useful for describing the status of the newborn at birth and his or her subsequent adaptation to the extrauterine environment, but it should not be used for the need for resuscitation or the steps to be taken. Resuscitation, when it is indicated, should be initiated before the 1-minute Apgar score is obtained. Apgar scores should be assigned at 1 minute and 5 minutes after birth, and if the 5-minute Apgar score is less than 7, additional scores should be assigned every 5 minutes for up to 20 minutes. The American Academy of Pediatrics (AAP) recommends the use of an assisted Apgar Scoring System that documents the assistance the infant is receiving at the time of assignment of the score. The Apgar score provides a convenient shorthand for reporting the status of the newborn infant and the response to resuscitation. It has been used inappropriately to predict specific neurologic outcome in the term infant. There are no consistent data on the significance of the Apgar score in preterm infants. The Apgar score has limitations, and it is inappropriate to use it alone to establish the diagnosis of asphyxia. An Apgar score assigned during resuscitation is not equivalent to a score assigned to a spontaneously breathing infant. An expanded Apgar score reporting form will account for concurrent resuscitation interventions and provide information to improve systems of perinatal and neonatal care. The purpose of this document is to place the Apgar score in its proper perspective. The Neonatal Resuscitation Program (NRP) guidelines, Apgar score and subsequent neurological dysfunctions are also discussed. The review also examines the occurrence of 5-minute Apgar score of 0 and seizures or serious neurologic dysfunctions. Perinatal asphyxia is a major cause of neurologic sequelae in term newborns. Apgar score is useful for conveying information about the newborn's overall status and response to resuscitation. However, resuscitation must be initiated, if needed, before the 1-minute score is assigned. Therefore, Apgar score is not used to determine whether the need for initial resuscitation steps are necessary, or when to use them. IntroductionIn 1952, Dr. Virginia Apgar devised a scoring system that was a rapid method of assessing the clinical status of the newborn infant at 1 minute of age and the need for prompt intervention to establish breathing (1). A second report evaluating a larger number of patients was published in 1958 (2). This scoring system provided a standardized assessment for infants after delivery. As soon as the baby is born a delivery nurse will set one timer for 1 minute and another for 5 minutes. When each of these time periods is up, a nurse or physician will give the baby her first "tests" called Apgars. It helps the physician estimate the baby's general condition at birth. The Apgar score is comprised of five areas:
Each of these areas is given a score of 0, 1, or 2 to quantify clinical abnormalities, depressed reflex response and apnea or gasping respirations. The expanded Apgar form is important because it allows documentation of the resuscitative efforts being done at the same time the score is assigned. Physicians need to recognize that an Apgar score should not be used to make predictions about an infants neurologic outcome, but that a low score increases the statistical risk of an abnormal outcome. There are many factors why an Apgar score by itself cannot be used to diagnose asphyxia, and many reasons other than asphyxia can result in low Apgar scores, such as, drugs, trauma, congenital anomalies, infections, hypoxia, hypovolemia, and preterm birth. A healthy preterm infant with no symptoms of asphyxia may receive a low score only because of immaturity. Therefore, it is important not to use the Apgar score alone to diagnose asphyxia. Physicians are encouraged to be consistent when assigning an Apgar score during resuscitation. The expanded Apgar score reporting form may be useful in determining whether continued resuscitative efforts are indicated, and it should continue to be used for any newborn receiving resuscitation.
Each characteristic is given an individual score; two points for each of the five categories if all is completely well; then all scores are totaled. For example, let's say the baby has a heart rate of more than 100, cries lustily, moves actively, grimaces and coughs in response to the syringe, but is blue; her 1-minute Apgar score would be 8 (2 points off because the baby is blue not pink). Most newborn infants have Apgar scores greater than 7. Because their hands and feet remain blue until they are quite warm, few score a perfect 10. The NRP current guidelines state that "if there is no heart rate after 10-minutes of complete and adequate resuscitation efforts, and there is no evidence of other causes of newborn compromise, discontinuation of resuscitation efforts may be appropriate. Current data indicate that, after 10 minutes of asystole, newborns are very unlikely to survive, or the rare survivor is likely to survive with severe disability" (3). Neonatal Encephalopathy and Neurologic Outcome, 2nd Edition, published in 2014 by the American College of Obstetricians and Gynecologists (ACOG) with the American Academy of Pediatrics (AAP), defines a 5-minute Apgar score of 7-10 as reassuring, a score of 4-6 as moderately abnormal, and a score of 0-3 as low in the term infant and late-preterm infant. (4). That document considers an Apgar score of 0-3 at 5-minutes or more as a non-specific sign of illness, which "may be one of the first indications of encephalopathy" (4). However, a persistently low Apgar score alone is not a specific indicator for intrapartum compromise. Further, although the score is used widely in outcome studies, its inappropriate use has led to an erroneous definition of asphyxia. Asphyxia is defined as the marked impairment of gas exchange leading, if prolonged, to progressive hypoxemia, hypercapnia, and significant metabolic acidosis. The term asphyxia, which describes a process of varying severity and duration rather than an end-point, should not be applied to birth events unless specific evidence of markedly impaired intrapartum or immediate postnatal gas exchange can be documented based on laboratory testing. Apgar Score and ResuscitationThe 5-minute Apgar score, and particularly a change in the score between 1 minute and 5 minutes, is a useful index of the response to resuscitation. If the Apgar score is less than 7 at 5-minutes, the NRP guidelines state that the assessment should be repeated every 5 minutes for up to 20 minutes. However, an Apgar score assigned during a resuscitation is not equivalent to a score assigned to a spontaneously breathing infant (5). There is no accepted standard for reporting an Apgar score in infants undergoing resuscitation after birth because many of the elements contributing to the score are altered by resuscitation. The concept of an assisted score that accounts for resuscitative interventions has been suggested, but the predictive reliability has not been studied. To correctly describe such infants and provide accurate documentation and data collection, an expanded Apgar score report form is encouraged (see below). This expanded Apgar score also may prove to be useful in the setting of delayed cord clamping, where the time of birth (complete delivery of the infant), the time of cord clamping, and the time of initiation of resuscitation all can be recorded in the comments box. Expanded Apgar Score Form: 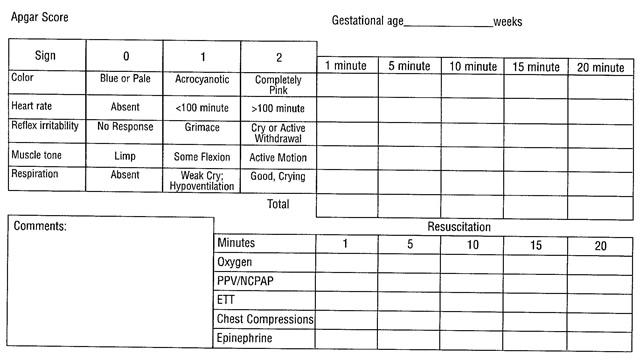 The Apgar score alone cannot be considered to be the evidence of or a consequence of asphyxia (6). Many other factors, including non-reassuring fetal heart rate monitoring patterns and abnormalities in umbilical arterial blood gases, clinical cerebral function, neuroimaging studies, neonatal electroencephalography, placental pathology, hematologic studies, and multisystem organ dysfunction need to be considered in diagnosing an intrapartum hypoxic-ischemic event. When a Category I (normal) or Category II (indeterminate) fetal heart rate tracing is associated with Apgar scores of 7 or higher at 5-minutes, a normal umbilical cord arterial blood pH (+/- 1 standard deviation), or both, it is not consistent with an acute hypoxic-ischemic event (7). Prediction of OutcomeA 1-minute Apgar score of 0-3 does not predict any individual infant's outcome. A 5-minute Apgar score of 0-3 correlates with neonatal mortality in large populations, but does not predict individual future neurologic dysfunction (8). Some studies stated that low Apgar scores at 5 minutes are associated with death or cerebral palsy, and this association increased if both 1- and 5-minute scores were low (9). An Apgar score at 5-minutes in term infants correlate poorly with future neurologic outcomes. For example, a score of 0-3 at 5-minutes was associated with a slightly increased risk of cerebral palsy compared with higher scores. Conversely, 75% of children with cerebral palsy had normal scores at 5 minutes (10). In addition, a low 5-minute score in combination with other markers of asphyxia may identify infants at risk of developing seizures (odd ratio: 39; 95% confidence interval; 3.9-392.5). The risk of poor neurologic outcomes increases when the Apgar score is 3 or less at 10, 15, and 20 minutes (11). A 5-minute Apgar score of 7 to 10 is considered normal. Scores of 4, 5, and 6 are intermediate and not markers of increased risk of neurologic dysfunction. Such scores may be the result of physiologic immaturity, maternal complications, and other factors. Because of these other conditions, the Apgar score alone cannot be considered evidence or a consequence of asphyxia. Other factors including non-reassuring fetal heart rate monitoring patterns and abnormalities in umbilical arterial blood gases, clinical cerebral function, neuroimaging studies, neonatal electroencephalography, placental pathology, hematologic studies, and multisystem organ dysfunction need to be considered when defining an intrapartum hypoxic-ischemic event as a cause of cerebral palsy. Attempts to Combine the Apgar with Umbilical Acid-Base AssessmentUmbilical cord blood gas analysis allows for an objective assessment of fetal acid-base status and determination of fetal well-being at the time of delivery. Current guidelines suggest that physicians should obtain both cord blood samples (arterial and venous) in all high-risk pregnancies and especially in cases of low 5-minute Apgar scores, severe fetal growth restriction, maternal thyroid disease, intrapartum fever, multifetal gestation, or abnormal fetal heart rate tracings (12). Furthermore, universal collection of cord blood gases at every delivery has been adopted at some institutions. As the limitations of the Apgar score in this regard became clear, there was hope that umbilical pH, a more objective if indirect measure of fetal oxygenation, would allow accurate prediction of permanent neurologic injury, either alone or in conjunction with the Apgar score. The reason some infants with normal pH may have a depressed Apgar score is clear when one considers the factors unrelated to asphyxia, such as: airway obstruction, airway suctioning, anterior horn cell disease, central nervous system (CNS) abnormalities, congenital infection, drugs/anesthesia, muscle disease, prematurity, primary cardiorespiratory disease and trauma. Despite their low Apgar scores, such infants are not at risk for neurologic sequelae due to asphyxia. Whenever low Apgar scores are assigned, a careful investigation for any of these factors should be undertaken. The converse of this situation, the finding of a low umbilical artery pH with a normal Apgar score, is often explained by rapid accumulation of CO2, the so-called fetal respiratory acidosis. Although pH is a log function, accumulation of CO2 due to interruption of gas exchange (e.g. from cord occlusion) results in a fall in pH that can be roughly estimated by the linear equation of a 10-mm increase in pCO2 with a 0.1 drop in pH. The rate of change of fetal pH due to this mechanism can be very rapid during labor. With repeated scalp sampling, it has been shown that a single deep variable deceleration can be associated with a rise in pCO2 of 10-mm. Such a change in pCO2 could result in a drop of pH from 7.25 to 7.15. Infants born with umbilical artery respiratory acidemia are not at marked risk for asphyxia neurologic sequelae. If umbilical pH is to be measured, the pCO2 should always be assessed as well. Less commonly, the finding of umbilical acidemia in a vigorous infant may be because of maternal acidemia on the fetal circulation (13). To assess the utility of umbilical cord venous blood gas measures for prediction of umbilical artery pH and base deficit acidemia, this study concluded that venous blood gas parameters are powerful predictors of arterial blood gas pH and base deficit and can be used to predict the likelihood of fetal acidemia when the cord arterial blood gas is not available (14). In order to determine the likelihood that an acute hypoxic-ischemia event that occurred within close temporal proximity to labor and delivery contributed to neonatal encephalopathy, it is recommended that a comprehensive multidimensional assessment be performed of neonatal status and all potential contributing factors, including maternal medical history, obstetric antecedents, intrapartum factors (including fetal heart rate monitoring results and issues relating to the delivery itself), and placental pathology. In confirmed cases of neonatal encephalopathy, the following assessment will determine the likelihood that an acute peripartum or intrapartum event was a contributor. This list is based on the premise that neonatal encephalopathy that is due to hypoxia-ischemia will be accompanied by abnormal neonatal signs and be associated with contributing events in close temporal proximity to labor and delivery. Neonatal Encephalopathy and Neurologic OutcomeNeonatal encephalopathy is a clinically defined syndrome of disturbed neurologic function in the earliest days of life in an infant born at or beyond 35 weeks of gestation, manifested by a subnormal level of consciousness or seizures, and often accompanied by difficulty with initiating and maintaining respiration and depression of tone and reflexes. This expanded clinical definition must be put into use based on measures that can be reliably and accurately implemented by trained staff. The first mandatory step in assessment of neonatal encephalopathy is to confirm whether a specific infant meets the case definition. The goal of the assessment is to compile a constellation of markers concerning neonatal status, contributing events, and developmental outcome to determine if they are consistent with acute hypoxia-ischemia and may not be explained by other etiologies. Thus, when more of the elements from each of the item categories are met, it becomes increasingly more likely that peripartum or intrapartum hypoxia-ischemia played a role in the pathogenesis of neonatal encephalopathy. Neonatal Signs Consistent with an Acute Peripartum or Intrapartum Event (13) A. Apgar Score of Less Than 5 at 5-minutes and 10-minutes
B. Fetal Umbilical Artery Acidemia
C. Neuroimaging Evidence of Acute Brain Injury seen on Brain Magnetic Resonance Imaging or Magnetic Resonance Spectroscopy with Hypoxia-Ischemia
D. Presence of Multisystem Organ Failure consistent with Hypoxic-Ischemic Encephalopathy
Type and Timing of Contributing Factors that are Consistent with an Acute Peripartum or Intrapartum Event (15) A. A Sentinel Hypoxic or Ischemic Event Occurring Immediately Before or During Labor and Delivery
B. Fetal Heart Rate Monitor Patterns Consistent with an Acute Peripartum or Intrapartum Event
C. Timing and Type of Brain Injury Patterns based on Imaging Studies consistent with an Etiology of and Acute Peripartum or Intrapartum Event
D. No Evidence of Other Proximal or Distal Factors that could be Contributing Factors Developmental Outcome is Spastic Quadriplegia or Dyskinetic Cerebral Palsy Neuroimaging Advances over the Past DecadeWith the wider use of MRI, the recognition of different patterns of injury has become established. Two main patterns often are distinguished on MRI: 1) the basal-ganglia-thalamus pattern, and 2) the watershed or border zone predominant pattern. In the interpretation of the literature on MRI in neonatal encephalopathy, there are two major weaknesses: 1) the exact timing of the insult is generally not known, and more importantly; 2) there are little to no data on the neuropathological correlate of the MRI pattern. MRI studies have defined that the vast majority of cases of cerebral injury that are seen in term-born infants with neonatal encephalopathy are acute. In comparison, epidemiologic studies have suggested that 70% of causation is related to chronic antenatal factors (13),(14). This apparent contradiction reflects the fact that the MRI studies relate imaging findings in the first 2-3 weeks of life and demonstrate a subacute pattern. These studies cannot, however, delineate if the injury occurred during labor or within the days before labor and delivery. There are few studies that have imaged infants in the first day of life to assist in the timing of ischemic cerebral injury. MRI can provide mutual information from diffusion-weighted imaging, conventional imaging, and magnetic resonance spectroscopy, which can inform timing. Information regarding the likely timing is best obtained with early imaging (first 24 - 96 hours of life) with further follow-up imaging to define the full nature of the abnormalities, optimally at 10 days of life (but with an acceptable window between 7 days and 21 days of life, depending on the logistics of acquiring MRI in the clinical setting). It is now accepted that identifying the predominant pattern of brain injury is an important predictor of neurodevelopmental outcome for a term newborn with encephalopathy. It is important to note that most studies that relate patterns of injury to neurodevelopmental outcome undertook imaging after day 7 of life. Conventional images provide a robust measure of the nature and severity of injury when performed after 1 week from the initial insult, which correlates well with neurodevelopmental outcome. Conventional MRI in the first 24 96 hours of life may underestimate the total extent of the injury but is better in timing. In summary, although MRI studies suggest that the period around the time of birth accounts for more than 75% of the causative period, studies have not systematically investigated the extent to which injury may have occurred during the 24 hours before delivery (16). Therefore, studies of early (first 48 hours of life) and serial (e.g. day 1, 4, 10 of life) MRI in term-born encephalopathic infants are needed and will assist in determining the evolution of imaging findings. These studies should include careful evaluation of the placenta. UPDATES on NEONATAL ENCEPHALOPATHY and CEREBRAL PALSYCerebral palsy is defined as the static neuromuscular disorder characterized by an abnormal control of movement or posture appearing early in life that is not the result of a recognized progressive disease (15). The prevalence of cerebral palsy is approximately 2 per 1,000 live births (17). Of these, a large proportion has no identifiable cause of brain impairment. Of those for whom asphyxia is likely to have played a role, differences in the timing of the asphyxia episode may alter the neonatal presentation and clinical course. For example, some neonates may have been injured early in gestation and yet survived in utero to be liveborn; others may have injured acutely intrapartum and may have died if not for intervention. In fact, the contribution of intrapartum events to the overall incidence of cerebral palsy attributable to "asphyxia" is relatively small and has been estimated to be approximately 10% of all incidences (18). Among neonates for whom asphyxia is the most likely cause of later cerebral palsy, the clinical presentation may be complicated by the timing, mechanism and severity of an asphyxia episode. The physiological mechanisms leading to asphyxia remain poorly understood. Because an asphyxia episode occurring intrapartum may be preventable, a variety of approaches has been used to ascertain the timing of this event. These approaches have largely depended on fetal monitoring characteristics, supplemented by adjunctive fetal and neonatal clinical observations (19). Criteria to define an acute intrapartum hypoxic event as sufficient to cause cerebral palsy:(20)
The criteria developed by the Task Force on Neonatal Encephalopathy and Cerebral Palsy appear to provide a template that may be useful for the analysis of cases involving intrapartum neonatal asphyxia injury. Whether this approach will also be valuable in populations of neonates with differing intrapartum fetal heart rate patterns remains to be investigated. Nevertheless, fetuses with a sudden, rapid, and sustained deterioration of the fetal heart rate during labor from a previously reactive fetal heart pattern who were found to have cerebral palsy appeared to demonstrate clinical characteristics that were consistent with the criteria recommended by the Task Force on Neonatal Encephalopathy and Cerebral Palsy. Low Apgar Scores and School PerformancePerinatal asphyxia is a major cause of neurologic sequelae in term newborns. The theory of a "continuum of reproductive casualty" implies that severe hypoxic events can cause death or obvious neurological deficit, but mild events might cause subtle defects in cognitive function that are only detectable as the child grows older (21). Mild hypoxic events are more common than severe events and do not usually lead to obvious neurological dysfunction. However, these events could lead to subtler cognitive impairment that could have a widespread effect on a larger population basis. No well-defined definition of perinatal asphyxia exists, and the definition has evolved from a single indicator definition to a multiple indicator definition (22). A low Apgar score has been considered a weak marker of asphyxia because of its low sensitivity and specificity, but few studies have addressed the long-term cognitive effects of a low Apgar score. In this study the aim was to elucidate the sole effect of an Apgar score less than 7 at 5 minutes after birth and cognitive performance as adolescents, as measured by school graduation grades from compulsory school at 16 years of age (23). The conclusion was an Apgar score of less than 7 at 5 minutes after birth is associated with subtle cognitive impairment, as measured by academic achievement at 16 years of age. This study has a long follow-up and addresses the long-term effects of a low 5-minute Apgar score. The limitations may be the maternal substance abuse, maternal medical conditions, and socioeconomic status, that cannot be adjusted for. However, the authors have adjusted for maternal smoking and maternal educational level. These results show a strong association between 5-minute Apgar scores and future cognitive function and are a useful tool in the assessment of obstetric care. PATIENT SAFETYEnhancing patient safety requires changing the culture of health care delivery from one that names and blames to one that is dedicated to reducing medical errors through a constructive, non-threatening, and professional process. The increased risk of 5-minute Apgar score of 0 and seizures or serious neurologic dysfunction of out-of-hospital births should be disclosed by obstetric practitioners to women who express an interest in out-of-hospital birth (24). Physicians should address patients' motivations for out-of-hospital delivery by continuously improving and compassionate care of pregnant, fetal, and neonatal patients in the hospital setting. The increased risks of home birth should be disclosed by all obstetric practitioners to all pregnant women who express an interest in out-of-hospital birth. Physicians also have a professional responsibility to address the root cause motivating patients to desire out-of-hospital birth by improving compassionate and safe hospital care and addressing concerns about interventions. SummaryThe Apgar score describes the condition of newborn infant immediately after birth, and when properly applied, it is a tool for standardized assessment. It also provides a mechanism to record fetal-to-neonatal transition. Apgar scores do not predict individual mortality or adverse neurologic outcome. However, based on population studies, Apgar scores of less than 5 at 5-minutes and 10-minutes clearly confer an increased relative risk of cerebral palsy, and the degree of abnormality correlates with the risk of cerebral palsy. Most infants with low Apgar score, however, will not develop cerebral palsy. The Apgar score is affected by many factors, including gestational age, maternal medications, resuscitation, and cardiorespiratory and neurologic conditions. If the Apgar score at 5- minutes is 7 or greater, it is unlikely that peripartum hypoxia-ischemia caused neonatal encephalopathy. Monitoring of low Apgar scores from a delivery service can be useful. Individual case reviews can identify needs for focused educational programs and improvement in systems of perinatal care. Analyzing trends allows for the assessment of the effect of quality improvement interventions. It has led to the understanding that an intrapartum asphyxia insult leading to permanent neurologic dysfunction is rare. Such an insult is characterized by profound, non-respiratory acidemia (umbilical artery pH <7.00). although there may be a prolonged depression of the Apgar score (0-3 for greater than 5 minutes), this is not a consistent finding. Most important, if the newborn does not show clear-cut evidence of encephalopathy in the nursery, there does not appear to be an increased risk of abnormal neurologic outcome, even in cases of low Apgar scores or umbilical acidemia (25). The advent of improved neonatal neuroimaging and magnetic resonance spectroscopy suggests that a significant number of infants with neonatal encephalopathy and subsequent cerebral palsy suffered causative intrapartum or peripartum events. Although the precise timing of such insults within 24 to 48 hours of delivery cannot be established from such contemporary imaging data, increased attention must be placed on optimizing intrapartum monitoring. Detection of hypoxic ischemic encephalopathy should prompt expeditious hypothermic therapy. Moreover, because evidence is strong that a focused patient safety program can reduce the occurrence of such adverse obstetrical outcomes and reduce malpractice claims, every obstetric unit in the United States should adopt such a program. References
|