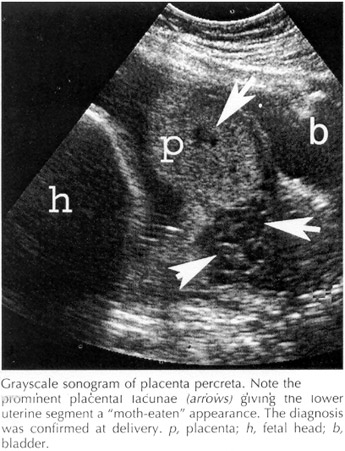
Placenta AccretaWHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC). The incidence of placenta accreta has increased 10 fold in the past 50 years and now occurs with a frequency of 1 per 2,500 deliveries. Women who have had 2 or more cesarean deliveries with anterior or central placenta previa have nearly a 40% risk of developing placenta accreta. An abnormally adherent placenta, although an uncommon condition, assumes considerable significance clinically because of morbidity and at times mortality from severe hemorrhage, uterine perforation, and infection. The incidence of placenta accreta, increta and percreta have increased because of the increased cesarean delivery rate. This document reflects emerging clinical and scientific advances on this subject. If the diagnosis or a strong suspicion is formed before delivery, the patient should be counseled about the likelihood of hysterectomy and blood transfusion. The term Placenta Accreta is used to describe any placental implantation in which there is abnormally firm adherence to the uterine wall. As the consequence of partial or total absence of the deciduas basalis and imperfect development of the fibrinoid layer, placental villi are attached to the myometrium. In Placenta Increta, it invades the myometrium and in Placenta Percreta, it penetrates through myometrium. The abnormal adherence may involve all of the cotyledons (total placenta accreta), a few to several cotyledons (partial placenta accreta), or a single cotyledons (focal placenta accreta). The histological diagnosis of accreta cannot be made from the placenta alone, the entire uterus or curettings with myometrium are necessary. Abnormal placental adherence is found when decidual formation is defective. The risk factors for placenta accreta include: In the presence of any of these conditions, the obstetric care provider must have a high clinical suspicion for placenta accreta and take appropriate precautions. Antepartum hemorrhage is common, but in the great majority of cases, bleeding before delivery is the consequence of coexisting placenta previa. Myometrial invasion by placental villi at the site of a previous cesarean section scar may lead to uterine rupture before labor. Massive hemo-peritoneum can also occur. In women whose pregnancies go to term, however, labor will most likely be normal in the absence of an associated placenta previa or an involved uterine scar. Early in pregnancy, the maternal serum alpha-fetoprotein level may be increased. The possibility exists that placenta increta might be diagnosed in antepartum. Ultrasonography may be helpful to make the diagnosis in the antepartum period. The lack of the usual sub-placental sonolucent space or hypo-echoic retro-placental zone, may be absent in the presence of placenta increta. Mounting evidence suggests that magnetic resonance imaging and color Doppler studies also may be useful to define an abnormally implanted placenta (1). At this time no diagnostic technique gives the clinicians 100% assurance of either ruling in or ruling out the presence of placenta accreta. The sonographic features suggestive of placenta accreta are: irregularly shaped placental lacunae (vascular spaces) within the placenta, thinning of the myometrium overlying the placenta, loss of the retroplacental "clear space", protrusion of the placenta into the bladder, increased vascularity of the uterine serosa-bladder interface, and on Doppler ultrasonography turbulent blood flow through the lacunae (3). Many studies have found at 15-20 weeks of gestation, that the presence of lacunae in the placenta is the most predictive sonographic sign of placenta accreta, with a sensitivity of 79% and a positive predictive value of 92%. These lacunae may give the placenta a "moth-eaten" or Swiss-cheese" appearance. Obliteration of the retroplacental "clear space", which is the finding most commonly thought to be associated with placenta accreta is only 57% sensitivity and false negative rate of 48.4%. After 20 weeks of gestation, the sensitivity of these findings increases with values of 93% and 80% for lacunae and obliteration of the retroplacental clear space, respectively. Comstock et al also found that a sonographic appearance of apparent bulging into the bladder may occur in cases of placenta accreta without increta or percreta. Power and color Doppler are often used for the diagnosis of placenta accreta, demonstrating turbulent flow through placental lacunae. However, in the majority of cases this imaging modality does not significantly improve the diagnosis over that achieved by grayscale sonography alone. Thus, in majority of clinical situations, Doppler should not be the primary technique used to diagnose placenta accreta (4). Although most studies have suggested reasonable diagnostic accuracy of MRI for placenta accreta, it appears that MRI is no more sensitive than ultrasonography for diagnosing placenta accreta (5). Ultrasonography is readily available in most centers, whereas MRI is costly and relatively inaccessible. Therefore, at the present time, sonography is the primary imaging modality for diagnosing placenta accreta. However, when there is a posterior placenta accreta, ultrasonography may be less than adequate, and MRI may be superior to ultrasonography for this specific indication. The problems associated with delivery of the placenta and subsequent developments vary appreciably, depending upon the site of implantation, depth of myometrial penetration, and number of cotyledons involved. With more extensive involvement, hemorrhage becomes profuse as delivery of the placenta is attempted. It is likely that focal placenta accreta with implantation in the upper uterine segment develops much more often than is recognized. The involved cotyledon is torn from the placenta and adheres to the implantation site with increased bleeding. This may be one mechanism for formation of placental polyps. With total placenta accreta, there may be very little or no bleeding, at least until manual placental removal is attempted. At times, traction on the umbilical cord will invert the uterus. The usual attempts at manual removal will not succeed because a cleavage plane between the maternal placental surface and the uterine wall cannot be developed. At the time of delivery blood transfusion, blood products and clotting factors should be available. Cell saver technology should be considered. If a strong suspicion is formed before delivery, the appropriate location and timing for delivery should be considered to allow access to adequate surgical personnel and equipment. A preoperative anesthesia assessment should be obtained. Profuse hemorrhage can occur when attempting to separate the placenta. The safest treatment in this circumstance is prompt hysterectomy. If the clinician is extremely confident in the diagnosis, it may be prudent to complete the delivery of the infant and proceed with hysterectomy while the placenta remains attached. Profuse hemorrhage after vaginal delivery also may be due to placenta accreta. In this instance, selective pelvic vessel embolization may be an alternative to hysterectomy or this procedure may decrease blood loss before or after hysterectomy. (2) Hysterectomy removes any prospect of future fertility and is associated with considerable morbidity and potential mortality, including that of surgical injury, given the distorted tissue planes and the need to operate in what is sometimes a blood-filled field. Conservative management has a limited role in carefully selected patients who desire future fertility. It has been suggested that delayed surgery leads to a less vascular surgical field and may have potential benefits when there is bladder involvement (6). Women offered conservative management should be counseled extensively that the outcomes are unpredictable and that there is a significant risk of serious complications including death. At the present time, this option cannot be recommended as a mainstay of therapy. The bladder is most frequently involved extrauterine organ when there is a placenta percreta. Management of the patient with bladder involvement requires careful perioperative planning and should involve urologist. Preoperative placement of uterine stents may aid in identification of the ureters, leading to a reduced risk of damage or injury to these structures. Involvement of bladder may require resection of bladder and occasionally of the ureters. Methotrexate, a folate antagonist, has been proposed as a conservative treatment for placenta accreta (7). Methotrexate acts primarily against rapidly dividing cells and therefore is effective against proliferating trophoblast. However, others have argued that, after delivery of the fetus, the placenta is no longer dividing and therefore methotrexate is of no value. The use of methotrexate did not prevent delayed hemorrhage. There are no large studies which have compared methotrexate with no methotrexate in the treatment of placenta accreta. Therefore, at the present time, there is no convincing data for or against the use of methotrexate for accreta. Placenta accreta has been reported to result in a 7% mortality rate as well as intraoperative and postoperative morbidity associated with massive blood transfusions, infection, ureteral damage, and fistula formation. In the past the most common form of "conservative" management was manual removal of as much placenta as possible and then packing of the uterus. Mortality rate was 25% with this type of management, thus the prompt hysterectomy is recommended in most of the cases. Recently, there has been heightened interest in clinical-pathologic correlation between placental abnormalities and adverse pregnancy outcomes. Continued research and education in this field should be encouraged. |