La santé des os: les stratégies de prévention de l'ostéoporoseBulletin WHEC pratique et directives cliniques de gestion pour les fournisseurs de soins de santé. Subvention à l'éducation fournie par la santé des femmes et Education Center (WHEC). Osteoporosis is an important health problem affecting mature women. It is a skeletal disorder characterized by loss of bone mass, deterioration of microarchitecture, and a decline in bone quality, all of which lead to an increased vulnerability to fracture. Many aspects of gynecology and obstetrics can affect bone health. Americans with osteoporosis or with low bone mass, approximately 80% are women. Osteoporosis-related fractures will occur in more than 40% of women over the age of 50. Hip fractures will occur in more than 40% of women over the age of 50. An estimated 1.3 to 1.5 million fractures occurring annually are attributed to osteoporosis, accounts for about 15% of the total. Within 1 year after a hip fracture, up to 20% of the victims will die, 25% of the survivors will be confined to long-term care facilities, and 50% will experience long-term loss of mobility. Spinal fractures can be associated with pain, loss of height, and deformities. Osteoporosis also is associated with tooth loss and the resorption of alveolar ridge. It is a potentially devastating and debilitating disease, and it is a public health threat in the United States. The fractures it causes have significant physical consequences, including limited mobility, deformity, and chronic pain, as well as functional limitations, such as a loss of independence. Fractures and osteoporosis also have serious psychosocial outcomes that can include anxiety, depression, loss of self-esteem, and the lack of rewarding social roles. The purpose of this document is to review the diagnosis, evaluation, and treatment of osteoporosis. Obstetricians and gynecologists play a major role in the prevention, diagnosis, and treatment of osteoporosis as outlined in this document. It is intended as an educational tool that presents current information. It is clear that age is one of most important factors related to bone quality, but exactly how age affects bone quality is not entirely clear. Changes in bone mass and microarchitecture have been well characterized. The other components that comprise bone quality are not well understood. Compliance (the consistency and accuracy with which a medication is initially followed) and persistency (the length of time a regimen is continued) are the most challenging aspects of osteoporosis management, both for patients who must take the medication and for physicians who treat this disorder. Because medication adherence remains an important issue in osteoporosis, education and attention to patient preferences for medications may help prevent the fractures that lead to the negative outcomes of osteoporosis. DefinitionThe World Health Organization (WHO) defines low bone density, or osteopenia as a bone mineral density between 1 and 2.5 standard deviations below the young adult mean and the osteoporosis be defined as a bone mineral density 2.5 standard deviations or more below the young adult peak mean. At the spine and hip, a 1 standard deviation decrease in bone mass is associated with approximately a two-fold increase in fracture risk (1). T score is the number of standard deviations above or below the mean average bone density value for young adult women. WHO provided criteria for diagnosing osteoporosis by dual-energy X-ray absorptiometry (DXA) scan, and this is the only test that has been validated for osteoporosis diagnosis. The most widely recommended method of diagnosing osteoporosis in the United States is bone densitometry. DXA of the lumbar spine and hip is the preferred method. Although peripheral sites such as the wrist and heel are predictive of osteoporosis and fractures, they are not suitable for monitoring treatment effects. Key Bone Density DefinitionsOsteoporosis A disease of the bone that is characterized by a predisposition to low-trauma fractures caused by low bone mass and disordered bone architecture. Defined by the WHO as a bone density T-score at or below -2.5 of normal peak bone density values for sex-matched young adults. A diagnosis of osteoporosis can also be made by proving the presence of a low-impact vertebral, hip, or wrist fracture. Low bone mass (previously osteopenia): Defined as a bone density T-score between -1 and -2.5 below the young adult mean. T-score: The difference in standard deviations between the value for the patient being tested and the mean value of a sex-matched group of adults aged 25 to 45 years. A T score of 0 indicates that the person being tested has a BMD that is at the mean for adults aged 25 to 45 years. Four standard deviations approximate the range of measurements from the 5th to 95th percentile. Z-score: The difference in standard deviations between the mean BMD of the patient being tested and a group of people of the same age and sex. A low Z-score may warrant an investigation for a secondary cause of osteoporosis, such as hypercalcemia, low 25-hydroxy vitamin D, or malabsorption syndrome. IntroductionThe main determinants of peak bone mass and bone quality are thought to be genetic (2). Many gene polymorphisms that affect bone quality have been identified. Other genetically linked findings such as changes in the Wnt signaling pathway, lipoprotein receptor-related protein 5 pathway, non-enzymatic glycation of collagen, and homocysteine levels also appear to contribute to the complexity of bone quality (2). Widely different age-adjusted hip fracture rates for women have been reported from around the world from 1 in 100,000 person-years among Nigerian black women to 421 in 100,000 person-years among Norwegian white women (3). Within the United States, there also is considerable variation in hip fracture rates, most notably between racial and ethnic groups. Caucasian women have the highest rates of hip fractures, and Mexican American women have rates in between the other two groups (4). These racial and ethnic differences are important in counseling and management because fracture rates do not always correlate with bone mineral density (BMD) across ethnic groups. For example, Chinese American women typically have lower areal BMD than Caucasian American women, but lower rates of hip and forearm fracture (4). It is postulated that greater cortical density and thicker trabeculae compensate for less trabeculae in smaller bones. Thus, both BMD and micro-architecture appear to play distinct roles in fracture vulnerability. However, apart from Chinese Americans, fracture rates correlated with BMD data collected in the National Health and Nutrition Examination Survey (NHANES) 2005-2006, which reported in African American women, and the lowest BMD in Caucasian American women (5). PathophysiologyThe functional part of bone is the remodeling unit. Bone can be divided into two major types: cortical and trabecular. Cortical bone forms the outer shell of all bones and accounts for 75% of total bone mass. Trabecular bone is the spongy, interlacing network of struts that forms the internal support within the cortical bone. Trabecular bone is concentrated in the vertebral bodies and pelvis and at the ends of the long bones. It accounts for 25% of total bone mass, but counts for most of the surface area of the bone. Bone remodeling units are limited to the bone surface. Because trabecular bone has large surface area, it has a higher turnover rate than cortical bone (6). The remodeling cycle can be conveniently divided into four phases: resting, resorption, reversal and formation. Each remodeling cycle may take several months to complete. During resting phase, stem-cells from the bone marrow are attracted to the bone surface and differentiate into osteoclasts. During the resorption phase, the osteoclasts remove bone using an acid pH to dissolve the minerals and proteolytic enzymes to digest the bone proteins. During reversal phase, the osteoclasts cease removing the bone and mesenchymal stem cells are attracted to the bone surface and differentiate into osteoblasts. During the formation phase, osteoblasts make new bone by first laying down a protein matrix (osteoid), which is then mineralized. Bone formation and resorption is an ongoing process that usually is balanced in young adults who have adequate nutrition and exercise and normal puberty. Bone mass peaks at approximately 30 years in both men and women. After reaching peak bone mass, about 0.4% of bone is lost per year in both sexes. In addition to this loss, women also lose approximately 2% of cortical and 5% of trabecular bone per year for the first 5 to 8 years following menopause (7). With aging, the coordinated balance between osteoclasts and osteoblasts may be disturbed, resulting in excessive bone loss. In women who are recently menopausal, excess bone loss is commonly due to excessive osteoblasts resorption. In later postmenopausal years, suppressed osteoblasts activity and inadequate formation of bone may play a major role in the progression of osteoporosis. 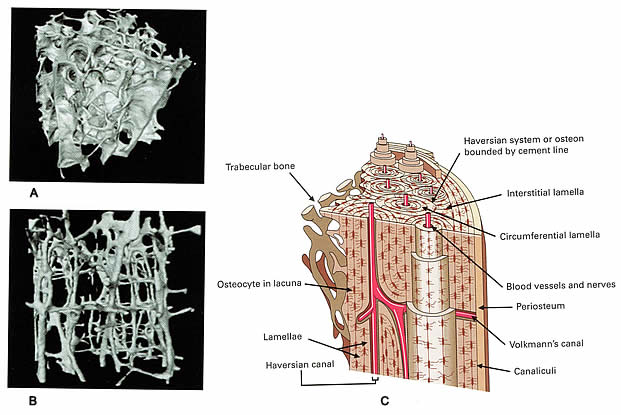 A. Normal bone; B. Clinically manifested osteoporosis bone; C. The microarchitecture of cortical bone Factors Affecting Bone MassBone mass is affected by these factors: family history, hormone levels, lifestyle and habits, nutrition, medications, and diseases that affect bone metabolism. Many studies have shown that the risk of osteoporosis is greater for white and Asian women than for African American women. Mexican American women have an intermediate risk. These racial differences are probably due, in part, to genetic determinants of body size, body composition, and bone metabolism. Genetic factors play a role in determining bone mass. Estradiol, testosterone, progesterone, cortisol, parathyroid hormone, thyroxine, growth hormone, and insulin all can influence bone mass. The mechanism by which estradiol regulates bone mass are not completely defined. Many lifestyle factors and habits influence bone mass. Cigarette smoking, excessive use of alcohol and high caffeine intake may be associated with decreased bone mass and an increased risk of hip fracture. Dietary calcium intake is an important modular of bone mass, especially during childhood, adolescence, and advanced age. Childhood calcium intake appears to influence adult hipbone mass (8). Vitamin D and its metabolites are essential to calcium metabolism and maintenance of mineral balance. In the United States, vitamin D deficiency rarely occurs except in select populations, such as those who are institutionalized with inadequate dietary intake and insufficient exposure to the sun. High protein diets acutely increase calcium excretion, but long-term, high protein diets are not associated with excess bone loss. Diets high in phosphorus can result in excess calcium loss, but daily phosphorus intake less than 2,00 mg is not harmful to bone (recommended daily allowance in menopausal women is 700 mg). Systemic medications such as glucocorticoids, thyroxine, and heparin can cause decreased bone mass. They have a direct effect on bone, causing inhibition of bone formation and enhancing bone resorption, and also decrease calcium absorption from the intestine and increase renal excretion of calcium. Hyperthyroidism is associated with decreased bone mass. Many metabolic bone diseases are associated with bone loss. These diseases often can be differentiated based on the measurement of serum calcium, phosphorus, and alkaline phosphatase. Osteoporosis can be caused by systemic diseases of the endocrine, hematopoietic, gastrointestinal and connective tissue systems. When osteoporosis is caused by systemic diseases, it is called secondary osteoporosis. Physical activity, adequate nutrition, and good health also are necessary for bone health (9). Exercise during the growth phase of life has the added advantage of modulating bone geometry in a way that enhances bone strength beyond what an increase in BMD alone can provide and may have longer lasting benefit. The most effective type of exercise to recommend has not yet been determined. However, a Cochorane analysis concludes that weight-bearing, resistance exercises and aerobics have a beneficial effect on spine BMD (no fracture data) and walking is beneficial for hip BMD (9). Risk Factors for Osteoporotic Fractures
DiagnosisDiagnosis needs to take into account other risk factors, including age, sex, white race, smoking and a family history of osteoporosis. There is no current accurate measurement of overall bone strength. Imaging technology is now available to determine bone mass with minimal radiation exposure, high accuracy, and high precision. DXA is preferred by most authorities. The precision of the measurement is approximately 1%. The radiation dose is less than 5 mrem. All modern DXA devices have an examination time in the range of 5 minutes. DXA instruments are available, which measure bone mass at peripheral sites such as in the forearm. Measurement of forearm, wrist, or hand bone density may not predict hip fractures as accurately as the direct measurement of hip-bone mass. One disadvantage of DXA is that bone spurs, aortic calcium, and arthritis may falsely elevate the reported bone density. Ultrasound bone mass measurement is a new technique that offers the potential advantage of avoiding exposure to ionizing radiation. Bone mass tests typically report three values:
In addition to bone density instruments, bone mass can be estimated through serial height measurements. Measurement of current height and comparison to reported maximal adult height can help establish the presumptive diagnosis of osteoporosis. Osteoporosis is likely to be present if the patient has lost more than 1 inch in height from her maximal adult height and the clinical setting is consistent with a diagnosis of osteoporosis. Biochemical markers can be useful to help identify women with high bone turnover and to monitor the effects of osteoporosis treatment. Markers that measure the rate of bone formation include serum bone alkaline phosphatase, serum osteocalcin, and serum procollagen I extension peptides. Markers that show the rate of bone resorption include urinary N-telopeptide, collagen crosslinks, urinary deoxypyridinoline, and urinary hydroxyproline (5). Other bone densitometry technologies can be used for predicting fracture risk, but the WHO classification does not apply to these systems. Peripheral DXA can be used at the heel, finger, or wrist. Quantitative ultrasound densitometry has no radiation exposure because the technology involves the use of either broadband ultrasound attenuation or speed of sound to derive a quantitative measurement of the bone. Common sites for measurement are the heel, patella, and tibia. Peripheral sites cannot be used to monitor treatment. Quantitative computed tomography can measure volumetric BMD of trabecular and cortical bone, either centrally at the spine and hip or peripherally at the forearm or tibia. There are substantially greater amounts of radiation exposure with this modality. Vertebral fracture assessment involves lateral spine imaging that can be performed by the lumbar spine DXA densitometer. A medical history of vertebral fracture is a reason to treat an at-risk woman even in the absence of a T-score of less than -2.5. A clinical diagnosis of osteoporosis can be established in the absence of imaging if there is a medical history of a low-trauma fracture in an at-risk woman (11). Low-trauma fractures are fractures that occur in a situation that would not be expected to cause fractures in most individuals (e.g. a vertebral fracture from opening a window or a simple fall from a standing position). These examples are in contradiction to a fracture caused by falling off a ladder or a fracture resulting from a car accident where higher trauma is present. Summary of Clinical Laboratory Data in Common Metabolic Diseases of the Bone
Bone Turnover MarkersBone turnover markers are byproducts of bone resorption (deoxypyridinoline, N-telopeptides, and C-telopeptides from the breakdown of type I collagen) and bone formation (osteocalcin, bone-specific alkaline phosphatase, and procollagen type I N-terminal propeptide associated with bone matrix synthesis). They can be measured in urine or serum to determine a high bone turnover state of high bone turnover, which may indicate higher risk of fracture. High levels reflect a state of high bone turnover, which may indicate higher risk of fracture. Bone turnover markers have been used in clinical trials of osteoporosis therapies to demonstrate group response to treatment. Changes in bone turnover markers occur earlier than changes in BMD and can be used to ascertain the effect of treatment more promptly. Use of bone turnover markers in the management of individuals is more challenging because levels vary from day to day and throughout a single day. Bone turnover markers cannot be used to diagnose osteoporosis, and the usefulness of markers as an incentive for adherence has been questioned (12). PreventionA healthy life-style is important for those at risk for osteoporosis. Cessation of smoking eliminates the effects of nicotine on estrogen binding. Moderation in alcohol and caffeine intake will improve general health, and weight-bearing exercises provide mechanical stimulation for bone remodeling. Sedentary life-style is associated with reduced bone mass. The benefits of physical exercise include maintenance of bone mass and an increase in muscle strength and coordination. The risk of falling increases substantially with aging. Most falls that result in hip fractures occur indoors. The living environment should be monitored to reduce the risk of falling. Prevention of osteoporosis begins in childhood and adequate calcium intake is important. Many children and adolescents do not achieve the recommended daily intake of calcium. Insufficient calcium intake also can accelerate age-associated bone loss. The two types of exercise necessary for bone health are weight-bearing and muscle-strengthening exercises. Weight-bearing exercises help build bone and keep bones strong. Strength training or resistance training helps to improve strength, balance, and flexibility. Perhaps its most important contribution is in preventing falls. Unfortunately, drawing evidence from research in this area is difficult for a variety of reasons. Until further research is conducted, knowledge about the impact of exercise on multiple aspects of osteoporosis is still insufficient. Institute of Medicine Recommended Dietary Allowance for Calcium and Vitamin D
TreatmentBefore initiating treatment, it is important to consider the possibility of secondary causes of osteoporosis. Fractures in a relatively young postmenopausal woman or a BMD lower than expected of age (e.g. a Z-score below normal for her age group) suggests the need to check for secondary causes of osteoporosis. An initial approach to evaluating for secondary causes of osteoporosis is suggested by American College of Obstetricians and Gynecologists (ACOG) are (13):
Treatments have been broadly classified as antiresorptive or anabolic, depending on the primary mechanism of action. There are many options for treating osteoporosis. BisphosphonatesThis class of drugs reduces bone resorption by inhibiting osteoclasts; slows bone loss and increases bone mass. Bisphosphonate are analogues of inorganic pyrophosphate, and a number of these are currently under development. Vertebral fractures are reduced by 48% in the women treated with alendronate and risedronate (14),(15). They are poorly absorbed from gastrointestinal tract and are recommended to be taken in the morning with 8 ounces of water, prior to any food or beverage. No food or beverage should be taken for the next 30 minutes to allow the alendronate to be absorbed. After taking alendronate or risedronate the woman should remain in an upright position, either sitting or standing to minimize the possibility of abdominal discomfort. The major side effects are abdominal discomfort, upper gastrointestinal bleeding, and musculoskeletal pain. One potential advantage of alendronate and risedronate are that they remain tightly bound to the surface of the bone for many years. It is not recommended for the patients with the renal failure or upper gastrointestinal problems. US Food and Drug Administration (FDA) has approved their use for the prevention of osteoporosis. The four bisphosphonates approved in the United States, alendronate, risedronate, ibandronate, and zoledronate, have been extensively studied in large randomized controlled trial that have demonstrated anti-fracture benefit. All bisphosphonates significantly reduce vertebral fractures by 32-65% (14),(15). Risedronate reduces non-vertebral fractures, and both alendronate and zoledronate significantly reduce hip fracture specifically. These drugs are classified as antiresorptive agents because the mechanism of action is inhibition of osteoclast resorption of bone. Inhibition of osteoclasts leads to a lesser decrease in bone formation by osteoblasts, but the net result is an increase in BMD and a decrease in bone turnover. Duration of effect after discontinuation may vary. Discontinuation of alendronate after 5 years of treatment resulted in maintenance of bone turnover markers below baseline for 5 years with BMD remained stable or decreased slowly (16). Discontinuing risedronate after 2 years of treatment resulted in significant loss of BMD during the first year (17). Zoledronate is contraindicated in patients with acute renal failure or creatinine clearance of less than or equal to 35 mL/min. Patients should be screened for renal disease before zoledronate infusion because renal failure has occurred after infusion in patients with compromised renal function. Caution with regard to renal function should be exercised with other drugs in this class as noted in the product information sheets. Hypocalcemia should be corrected before the use of these drugs. Government Approved Bisphosphonates for Postmenopausal Osteoporosis
Adverse effects of bisphosphonates include musculoskeletal aches and pains, gastrointestinal irritation, and esophageal ulceration. Potential risks reported after marketing include osteonecrosis of the jaw, seizures, atypical fractures of the femoral shaft, and esophageal cancer. A precise understanding of the true risk of these events has been difficult to determine because of the lack of data on the incidence of these problems in the general population. Although rare cases of osteonecrosis of the jaw have been reported in patients using bisphosphonates for osteoporosis (18). It has been seen most commonly after dental extractions in those being treated with large intravenous doses of bisphosphonates in association with supportive cancer therapy (19). There is no requirement to discontinue bisphosphonates for dental procedures. However, there is likely to be no harm in discontinuing a bisphosphonate temporarily for a dental procedure, if the patient so desires, given the long duration of action of bisphosphonates DenosumabDenosumab (Prolia), an antiresorptive treatment, is a human monoclonal antibody to the receptor activator of nuclear factor-kB ligand. The receptor activator of nuclear factor-kB ligand binds to the receptor activator of nuclear factor-kB on the surface of osteoclasts and promotes proliferation and differentiation of these osteoclasts. The antibody blocks this interaction therein decreasing bone resorption and increasing BMD as a result (20). Denosumab was approved in 2010 for treatment of postmenopausal women with osteoporosis who are at high risk of fracture. Studies revealed a vertebral and hip fracture reduction of 68% and 40%, respectively (20). Denosumab (Prolia) is administered subcutaneously every 6 months. A higher rate of infections that required hospitalization was seen in the initial trials. However, concerns about suppression of the immune system leading to increased rates of cancer were not substantiated. Partial Estrogen Agonists and AntagonistsRaloxifene (Evista) was the first drug in the class of partial estrogen agonists and antagonists (also known as selective estrogen receptor modulators) approved for the prevention and treatment of osteoporosis. It works as weak estrogen agonists in some systems, and estrogen antagonists in others. Raloxifene decreases the number and activity of osteoclasts and is approved for the prevention and treatment of postmenopausal osteoporosis. It decreases the incidence of vertebral fractures by 30% to 50% and increases BMD in the spine and femoral neck (21). Raloxifene is associated with a reduced risk of estrogen-dependent breast cancer, and may lower the risk of heart disease and stroke in women at high risk. However, the long-term effects of raloxifene on the vascular system are unknown, and increased risk of thromboembolic events similar to that reported with estrogens has been observed. As a result of its tendency to cause or exacerbate hot flashes, raloxifene is not recommended for newly menopausal women or for the treatment of vasomotor symptoms. However, it can reduce the risk of breast cancer in postmenopausal women at low risk of the disease, and is currently being evaluated for use in reducing the incidence of breast cancer in women at increased risk. CalcitoninCalcitonin (Miacalcin, Fortical) is a synthetic polypeptide based on salmon calcitonin, is available in a subcutaneous injectable form and as nasal spray. It inhibits bone resorption and is approved for the treatment of osteoporosis in women who are postmenopausal for at least 5 years. It increases vertebral bone mass and significantly reduces new vertebral fractures. It has been shown to reduce vertebral fractures and to reduce bone pain associated with vertebral fractures (22). Objection to injectable now can be avoided by intranasal spray. Risks are: development of neutralizing antibodies-effect and absorption is variable. One spray in one nostril delivers the recommended dose of 200 IU of salmon calcitonin. Fracture reduction is less robust than with other agents and was not seen in early postmenopausal women. It should not be used until women are 5 years from menopause. Adverse effects include flushing and nausea with subcutaneous injection and local irritation with nasal spray. It was one of first treatments for osteoporosis. It was widely used and showed effectiveness by increasing bone density at the spine and hip and by reducing hip, vertebral, and non-vertebral fractures. Estrogen / hormone therapy was also used to manage menopausal symptoms, and was widely used prior to the release of Women's Health Initiative (WHI) study findings in 2002. This trial found that estrogen / hormone therapy was effective at reducing hip fractures and preventing colon cancer, but that unexpectedly, women taking estrogen or estrogen plus progestin were found to have statistically significantly higher rates of cardiovascular disease, stroke, dementia, breast cancer, and other conditions that were studied. The investigators concluded that health risks associated with estrogen / hormone therapy outweighed the benefits (23). As a result of these findings, estrogen's status was changed by the FDA. It is currently approved for the prevention of osteoporosis in women who are also experiencing menopausal symptoms. In cases where women do not have such symptoms, they should use another agent for the prevention of osteoporosis. Contraindications are: undiagnosed abnormal genital bleeding; known, suspected, or history of cancer of the breast except in appropriately selected patients being treated for metastatic disease; known or suspected estrogen-dependent neoplasia; active deep vein thrombosis, pulmonary embolism, or a history of these conditions; active or recent (within the past year) arterial thromboembolic disease (for example, stroke, myocardial infarction); liver dysfunction or disease; known thrombophilic disorders (e.g. protein C, protein S, or antithrombin deficiency); known hypersensitivity to any of the ingredients in these products; and known or suspected pregnancy. It is an anabolic, approved for the treatment of severe osteoporosis in postmenopausal women that showed very robust fracture reduction at vertebral and non-vertebral sites in clinical trials (24). This drug has black-boxed warning because of the preclinical incidence of osteosarcoma in rats, although no increased osteosarcoma incidence has been reported in post-marketing surveillance of patients who received this agent. It is administered as a subcutaneous injection 20-microgram dose daily in conjunction with calcium and vitamin D for 24 months. After treatment period is completed, the patient may have to be maintained on another therapy, since the benefit of teriparatide wanes quite rapidly within 6 months to 1 year following discontinuation. Teriparatide has been studied in nursing home patients and was found to be safe and effective. However, there are few data suggesting it is actually being used, perhaps because of the need for a daily injection. Newer drugs for the treatment of osteoporosis have been evaluated in recent clinical trials. Odanacatib, an inhibitor of cathepsin K ( a proteinase believed to play a vital role in bone resorption and remodeling), decreased bone resorption, maintained bone formation, and increased BMD in a randomized, double-blinded, 2-year trial of postmenopausal women (25). AMG785 is a monoclonal antibody targeted against sclerostin, an osteocyte-secreted protein that negatively regulates osteoblasts and inhibits bone formation. In a randomized, double-blind, placebo-controlled trial in healthy men and postmenopausal women AMG785 demonstrated dose-related increase in bone-formation markers and significant increases in BMD (25). All major guidelines state that DXA screening should begin at age 65 years for women (26). Most guidelines also agree that DXA screening can be used selectively for women younger than 65 years if they are postmenopausal and have other risk factors for fracture. Bone density should be screened in postmenopausal women younger than 65 years if any of the following risk factors are noted: After treatment initiation, one DXA scan 1 year or 2 years later can be used to assess the effect of treatment. If the BMD is improved or stable (no significant change), the DXA does not usually need to be repeated in the absence of new risk factors (27). Testing generally should not be undertaken before 2 years after initiation of treatment because if often takes 18-24 months to document a clinically meaningful change. Although the use of depot medroxyprogesterone acetate (DMPA) is associated with loss of BMD, current evidence suggests that partial or full recovery of BMD occurs at the spine and at least partial recovery occurs at the hip after discontinuation of DMPA. Practitioners should not perform BMD monitoring solely in response to DMPA use because any observed short-term loss in BMD associated with DMPA use may be recovered and is unlikely to place an adolescent or adult woman at risk of fracture during use or in later years. Lower bone density T-scores generally indicate more severe osteoporosis and higher risk of fracture. Every decrease of 1 standard deviation from age-adjusted bone density represents approximately a 10-12% change in BMD and an increase in the risk of fracture by a factor of approximately 2. Although few would withhold treatment from a woman with osteoporosis (T-score less than -2.5), whether to treat a woman with higher bone density scores has become a subject for debate. The National Osteoporosis Foundation (NOF) has chosen a T-score of -2 for women without risk factors and -1.5 for women with additional risk factors as the threshold for therapeutic treatment (11). The Z-score may help in determining an alternative strategy, particularly with the Z-score is less than -1 and the patient would not have qualified for therapy solely on the criteria discussed above. Under these circumstances, a secondary cause for the bone loss should be sought. Monitoring a patient's response to treatment requires central bone densitometry. For women receiving osteoporosis therapy, bone mineral density BMD monitoring before 2 years of therapy are completed does not provide clinically useful information and may lead to erroneous assumption about the effect of therapy. Not observing an increase in BMD is not evidence of treatment failure. A decrease in vertebral bone mineral density greater than 4-5% indicates a need to evaluate the patient's compliance with therapy and dosing instructions and to search causes of bone loss. Follow-up and Long-term management for at least 5 years: The most widely used bone medications are the bisphosphonates -- alendronate, risedronate, ibandronate, and zoledronate. Other FDA-approved bone medicines include estrogen or estrogen-progestin hormone therapy, raloxifene, parathyroid hormone, and calcitonin. After initiating a bone medicine, a DXA BMD can be measured in 1 to 2 years to assess changes in bone density. If the bone density is stable or increases, continue therapy. If compliance with treatment has been good and bone density was demonstrated to significantly decrease with treatment, consider a new regimen and reassess bone density in 1 to 2 years. An alternative approach is to measure fasting urinary cross-linked N-telopeptides of type I collagen (NTX) or serum type I collagen C-telopeptides (CTX) before and 3 to 6 months after starting antiresorptive treatment, such as a bisphosphonate. If the marker has decreased by 50% or more, treatment is likely to be successful and bone density can be assessed in 2 years. If the marker has not decreased by 50%, non-compliance or incorrect dosing, such as taking the bisphosphonate with food, is likely causes of a poor marker response (12). All major guidelines state that treatment should be recommended for women who have a BMD T-score of less than or equal to -2.5. For women in the low bone mass category (T-score between -1 and -2.5), the fracture risk assessment tool (FRAX) calculator can be used to make an informed treatment decision. Women who are found to have a 10-year risk of major osteoporotic fracture greater than or equal to 20% or a risk of hip fracture greater than or equal to 3% using the FRAX calculator are candidates for medical pharmacologic therapy (28). Women who have had a low-trauma fracture (especially of the vertebra or hip) also are candidates for treatment even in the absence of osteoporosis on the DXA report. Women with osteoporosis or who are at risk of osteoporosis should be counseled about lifestyle changes to reduce the risk of bone loss and osteoporotic factors. Recommendations include performing weight-bearing exercises and muscle-strengthening exercises to reduce the risk of fractures and falls, taking the appropriate amount to vitamin D and calcium, stopping smoking and avoiding second-hand smoke, reducing alcohol intake, and adopting fall-prevention strategies. Treatment Selection: Bisphosphonates are generally considered first-line therapy, but raloxifene can be a good first choice in younger postmenopausal women for the previously described reasons. A woman using raloxifene can be transitioned to another therapy, such as bisphosphonate, in her 60s. Bisphosphonates can be selected on the basis of patient preference for a certain route of delivery or insurance coverage. Patients who have gastrointestinal problems or had gastrointestinal adverse effects with oral preparations may be better served with a parenteral bisphosphonate. For patients with adherence issues, an annual infusion of zoledronic acid may be preferred. Denosumab also is a good treatment option for the woman with a high risk of fracture. The subcutaneous injections every 6 months may appeal to some patients and not others. Combination therapy is not usually recommended. Although it may increase BMD to some degree, fracture data are not available. Risks, adverse effects, and cost with the use of two drugs may be higher than with one drug. Follow-up with DXA Scan: If the initial BMD report indicates low bone mass, FRAX should be used to determine if the woman has a high risk of fracture. If she does, treatment should be recommended. If FRAX does not indicate a high risk of fracture, data from the Study of Osteoporotic Fractures suggest a screening interval of 1-5 years for a woman older than 65 years with a normal BMD or mild bone loss (T-score greater than or equal to -1.5), a 5-year screening interval for a T-score from -1.5 to -1.99, and a 1-year screening interval for a T-score between Lighting - Provide accessible lighting for each part of the home use; use night-lights. Floors - Remove throw rugs or secure them to the floor; remove all clutter from the floor; move cords and cables away from traffic path; use non-skid wax. Storage - Store items at a height that does not require a step stool to reach. Bathroom - Install safety grab bars in bath and shower; apply non-skid strips to bath and shower floor. Indoors and Outdoor Stairs - Install handrails for the entire length of the stairs; provide adequate lighting; add non-skid treads or secure carpet indoors. Adherence has two components: compliance in taking the medication properly, and persistence in taking the medication over the long term. The consequences of poor adherence are highly detrimental in osteoporosis management; conversely, adherent patients have been shown to experience a 16% lower fracture rate than non-adherent patients (32). With regard to dosing schedule, weekly and monthly bisphosphonate use has demonstrated significantly longer persistence than daily use, and more weekly and monthly users met predetermined criteria for good compliance than did daily users. Polypharmacy for multiple chronic conditions in older patients substantially challenges adherence to osteoporosis treatment regimens. Drug-related adverse events in this population may also exacerbate existing comorbidities, further decreasing adherence. It has been found that one year after initiating treatment, 45% of patients stop filling their osteoporosis prescriptions; older age and increasing numbers of comorbid conditions and medications unrelated to osteoporosis treatment all predicted lower adherence (33). Nine FDA-approved pharmaceutical therapies currently exist for osteoporosis, an important increase since 1995. However, regardless of the type or efficacy of the medication, it can only work if patient takes it. Unfortunately, many patients do not regard osteoporosis as a serious or chronic disease and do not take their medication as prescribed (30). Compliance (the consistency and accuracy with which a medication is initially followed) and persistence (the length of time a regimen is continued) are the most challenging aspects of osteoporosis management, both for patients who must take the medication and for physicians who treat this disorder (31). Noncompliance to osteoporosis medication ultimately undermines the reason for taking medication and leads to the outcome of fracture. Indeed, a positive correlation exists between compliance to osteoporosis medication and lower fracture risk, as evidenced by a study of more than 11,000 postmenopausal women with osteoporosis. A 16% lower fracture rate occurred in adherent women who took at least 80% of the doses of their osteoporosis medications compared with non-compliant women (32). One way in which patients do not comply with medication prescriptions is called secondary non-adherence. In this case, patients fill their prescriptions, take them home, and start to take them. Then something happens (e.g. side effects, cost, and forgetfulness) and the patients are no longer following their prescribed medication routine. Many studies have examined secondary non-adherence in osteoporosis and interventions have been developed to improve patient medication behavior, although many have been ineffective (34). What can be done to improve medication behaviors in osteoporosis? Greater communication between patients and healthcare providers can improve compliance and persistence, which can be accomplished through e-mails and phone calls, in addition to regular office visits. Consistent follow-up with the patient through visits, e-mails, and phone calls is essential for assessment of treatment tolerance and adherence. In a study that examined the effect of nurse monitoring on adherence to osteoporosis therapy in postmenopausal women, monitoring of patients increased adherence to therapy by 57% at 1 year (34). Physicians who work with their patients to identify and resolve barriers that contribute to non-adherence, and who educate and empower patients in self-managing their medications, may help improve overall treatment adherence and facilitate positive treatment outcomes. Osteoporosis is a serious disease with a prevalence that is expected to increase as the population ages. This disease deserves attention of both patients and physicians, especially since very effective means exist for identifying and assessing patients at risk for osteoporosis. Advances in imaging technology allow clinicians to diagnose osteoporosis before a clinically significant fracture occurs. Numerous therapies are available for the prevention and treatment of osteoporosis in postmenopausal women; however, variations exist in the overall efficacy of these product types, and in the range of skeletal sites at which beneficial effects are observed. Women with low bone density or established osteoporosis should be offered estrogen replacement therapy, alendronate, or calcitonin therapy. Application of these new tools and treatment by obstetrician-gynecologist may reduce the number of osteoporotic fractures experienced by our aging population. It is not only in the area of pharmacotherapies that changes are anticipated within the next few years. Greater emphasis will almost certainly be placed on identification of the determinants of osteoporotic fracture risk, to enable more effective targeting of preventive measures to those women at greatest risk. Treatment should be recommended for: women with a T-score of -2.5 or less; women who have had low-trauma fracture; and women who have a T-score from -1 to -2.5 and a FRAX score greater than or equal to 20% for risk of a major osteoporotic fracture (defined as forearm, hip, shoulder, or clinical spine fracture) or both in the next 10 years. FDA approved therapies should be used for medical treatment: raloxifene, bisphosphonates, PTH, Denosumab, and calcitonin. What can be done to improve medication behaviors in osteoporosis? Greater communication between patients and healthcare providers can improve compliance and persistence, which can be accomplished through e-mails and phone calls, in addition to regular office visits. Consistent follow-up with the patient through visits, e-mails, and phone calls is essential for assessment of treatment tolerance and adherence. Physicians who work with their patients to identify and resolve barriers that contribute to non-adherence, and who educate and empower patients in self-managing their medications, may help improve overall treatment adherence and facilitate treatment outcomes. A number of safe and effective therapies, which can reduce fractures by approximately 50% at all sites, are available to treat osteoporosis. It is incumbent on physicians to identify at-risk individuals and implement appropriate therapy. Because medications adherence remains an important issue in osteoporosis, education and attention to patient preferences for medications may help prevent the fractures that lead to the negative outcomes of osteoporosis. |