Inherited Thrombophilias in Pregnancy
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC).
Identification of inherited thrombophilias has increased our understanding of one potential etiology for venous thromboembolism (VTE) and of hypercoagulability in general. Some contributing mutations, including the factor V Leiden mutation, prothrombin G20210A mutation, and the methylene tetrahydrofolate reductase (MTHFR) C677T polymorphism, are quite common in the general population. Over the past 10 years, some studies have suggested that inherited thrombophilia may be associated with preeclampsia and other adverse outcomes in pregnancy. There is limited evidence to guide screening for and management of these conditions in pregnancy. Successful pregnancy requires the avoidance of hemorrhage during implantation, endovascular cytotrophoblast remodeling of maternal spiral arteries, and during the third stage of labor, yet also requires the maintenance of a fluid uteroplacental circulation. Maintaining hemostatic balance during pregnancy requires alterations in both local uterine and systemic clotting, as well as anticoagulant and fibrinolytic proteins. There is a strong association between inherited thrombophilias and venous thromboembolism, which makes detection of these mutations a logical target for prevention strategies. Thrombosis is hypothesized to be the more common mechanism underlying cerebral palsy in many cases of maternal or fetal thrombophilia; for that reason, understanding the impact of maternal and fetal thrombophilia on pregnancy outcome is of paramount importance when counseling patients.
The purpose of this document is to review common thrombophilias and their association with maternal venous thromboembolism risk and adverse pregnancy outcomes, indications for screening to detect these conditions, and management options in pregnancy. Is a maternal and fetal thrombophilia work-up needed in women who give birth to a term infant with cerebral palsy? Prospective studies are needed to evaluate whether that is the case. In this review, the literature on fetal thrombophilia and its role in explaining some cases of perinatal stroke that lead, ultimately, to cerebral palsy are also discussed.
Background
Pregnancy is marked by increased clotting potential, decreased anticoagulant activity, and decreased fibrinolysis (1). The thrombotic potential of pregnancy is exacerbated by venous stasis in the lower extremities due to compression of the inferior vena cava and pelvic veins by the enlarging uterus, a hormone-mediated increase in venous capacitance, insulin resistance, and hyperlipidemia. Thus, it is not surprising that venous thromboembolism complicates approximately 1 in 1,600 births and is a leading cause of maternal morbidity in the United States (2). Prevalence of common inherited thrombophilias is:
- Factor V Leiden: Its prevalence in European populations is approximately 5% (3). Although the mutation is virtually absent in black Africans, Chinese, Japanese, and other Asian populations, it is present in 3% of African Americans whose ancestors are not recent immigrants. The mutation renders factor V refractory to proteolysis by activated protein C. Women who are heterozygous for factor V Leiden have been observed to account for approximately 40% of cases of venous thromboembolism during pregnancy; however, the risk of venous thromboembolism among pregnant women who are heterozygous for factor V Leiden without a personal history of venous thromboembolism or an affected first-degree relative with a thrombotic episode before age 50 years is less than 0.3% (4). In contrast, this risk increases to at least 10% among pregnant women with a personal or family history of venous thromboembolism (4). Pregnant women who are homozygous for factor V Leiden without a personal history of venous thromboembolism or an affected first-degree relative have a 1-2% risk for venous thromboembolism, whereas those with such a history have a 17% risk (4).
- Prothrombin G20210A: It is a point mutation that results in elevated circulating prothrombin levels (3). The prothrombin G20210A mutation is present in approximately 3% of the European population, and it has been reported to account for 17% of cases of venous thromboembolism in pregnancy (5). As with factor V Leiden, a personal history or history of venous thromboembolism in a first-degree relative before age 50 years increases the risk of venous thromboembolism in pregnancy. Without such a history, carriers of the prothrombin G20210A mutation have a less than 0.5% risk of venous thromboembolism during pregnancy; for a carrier with such a history, the risk exceeds 10% (5). Pregnant women who are homozygous for prothrombin G20210A mutation without a personal or positive family history have a 2-3% risk of venous thromboembolism in pregnancy, whereas such a history confers a substantially greater risk. The combination of factor V Leiden and prothrombin G20210A mutations has synergistic hypercoagulable effects. Those who are heterozygous for this combination, although present in only 1 per 10,000 patients, have a 4-5% risk of venous thromboembolism even without a personal family history (4),(5).
- Protein C Deficiency: It has been linked to more than 160 distinct mutations that produce a highly variable phenotype (3). The prevalence of protein C deficiency is 0.2-0.3% when determined by a functional assay with a cutoff of 50-60%. The risk of venous thromboembolism in pregnancy among the typical protein C deficient patient with a personal or family history has been reported to be 2-7% (6). Although rare, newborns homozygous for protein C deficiency will develop neonatal purpura fulminans and require lifetime anticoagulation (6).
- Protein S Deficiency: It generally has two causes, a silenced gene, or a mutation, which results in reduced free protein S antigen levels and activity (3). Detection of protein S deficiency using activity assays alone is subject to substantial variability due to fluctuating levels of protein S binding protein in pregnancy (7). Therefore, screening in pregnancy is necessary, cutoff values for free protein S antigen levels in the second and third trimesters have been identified at less than 30% and less than 24%, respectively (1). Among those with a positive family history, the risk of venous thromboembolism in pregnancy has been reported to be 6 to 7% (7). As with protein C deficiency, homozygous protein S deficiency results in neonatal purpura fulminans (6).
- Antithrombin Deficiency: It is highly thrombogenic but rare, the more than 250 associated mutations can decrease gene transcription, leading to reductions in both antigen and activity, or alter structure and function leading to normal antigen levels but decreased activity (8). The very rare homozygous state is associated with little or no antithrombin activity. The prevalence of antithrombin deficiency is approximately 1 per 2,500 patients (8). In nonpregnant patients, the risk of venous thromboembolism among antithrombin-deficient patients is increased more than 25-fold. Pregnancy may increase the thrombogenic potential of antithrombin deficiency substantially (8). However, this risk may be much lower in the absence of a positive personal or family history.
- Methylenetetrahydrofolate Reductase (MTHFR): Homozygosity for this gene mutation is the most common cause of hyperhomocysteinemia. Homozygosity for the MTHFR C677T and A1298C polymorphisms is present in 10-16% and 4-6% of all Europeans, respectively (9). However, MTHFR mutations by themselves do not appear to convey an increased risk for venous thromboembolism in either nonpregnant or pregnant women (9). Although hyperhomocysteinemia was previously reported to be a modest risk factor of venous thromboembolism, recent data indicate that elevated homocysteine levels are a weak risk factor of venous thromboembolism (10). This observation may reflect the folate-replete diet of developed nations, including folate supplementation studies with vitamin B supplementation in nonpregnant patients show no reduction in venous thromboembolism. Thus, there is insufficient evidence to support assessment of MTHFR polymorphisms or measurement of fasting homocysteine levels in the evaluation of a thrombophilic etiology for venous thromboembolism, and therefore, it is not recommended.
- Other Thrombophilias: A variety of other thrombophilias have been described, including alternative mutations in the factor V gene, a promoter mutation in the PAI-1 gene, protein Z deficiency, and activity-enhancing mutations in various clotting factor genes. Although they appear to exert little independent risk of venous thromboembolism, they may exacerbate risk among patients with the aforementioned mutations. However, there is insufficient evidence to recommend screening for these thrombophilias.
Inherited Thrombophilias and Adverse Pregnancy Outcomes
The role that inherited or acquired thrombophilias may play in pathogenesis of preeclampsia has been thoroughly investigated for several years. Data from this study (11) demonstrate that thrombophilia is associated with more than a 2.5-fold increase in risk of recurrence of preeclampsia. The results were similar in the subset of women with factor V Leiden and factor II mutations, the most common form of heritable thrombophilia. The observation that patients who have previously had preeclampsia are at increased risk for subsequent development of thromboembolic episodes is further epidemiologic evidence linking thrombophilia and preeclampsia. Factor V Leiden and factor II mutations and thrombophilia itself increase the risk for recurrence of preeclampsia; and women who have a history of preeclampsia should be screened for thrombophilia to identify a high-risk group of women who may be eligible for intervention trials (11). Meta-analyses and a retrospective cohort study have revealed an association between inherited thrombophilias and first-trimester pregnancy loss (12). The Eunice Kennedy Shriver National Institute of Child Health and Human Developments Maternal-Fetal Medicine Units Network tested low-risk women with a singleton pregnancy less than 14 weeks gestation. The Maternal-Fetal Medicine Units Network identified 134 women who were heterozygous for factor V Leiden among 4,885 pregnant women, and found no increase in the incidence of fetal loss (13). Similar findings of no increased risk of fetal loss were noted for maternal carriers of the prothrombin G20210A gene mutation (13).
Multiple case-control, cohort, and systematic review studies have failed to detect a significant association between factor V Leiden and intrauterine growth restriction (IUGR) less than the 10th percentile or less than the 5th percentile (14). A similar lack of association was noted between prothrombin G20210A mutation and IUGR (14). Overall, there is insufficient evidence to establish a link between thrombophilia and placental abruption. Prospective cohort analyses of factor V Leiden, prothrombin G20210A, and pregnancy outcome found no association with placental abruption (15). The Hordaland Homocysteine Study found an association between placental abruption and hyperhomocysteinemia greater than 15 micromol/L, but minimal association between homozygosity for the C677T MTHFR polymorphism and placental abruption (15).
Clinical Considerations
Screening for thrombophilias in controversial. It is useful only when results will affect management decisions, and is not useful in situations where treatment is indicated for other risk factors. Screening may be considered in the following clinical settings:
- A personal history of venous thromboembolism that was associated with non-recurrent risk factor (e.g., fractures, surgery, and prolonged immobilization). The recurrence risk among untreated pregnant women with such a history and a thrombophilia was 16% (16).
- A first-degree relative (e.g., parent or sibling) with a history of high-risk thrombophilia or venous thromboembolism before age 50 years in the absence of other risk factors in as much as affected women should receive prophylaxis.
In other situations, thrombophilia testing is not routinely recommended. Testing for inherited thrombophilias in women who have experienced recurrent fetal loss or placental abruption is not recommended. Although there may be an association in these cases, there is insufficient clinical evidence that antepartum prophylaxis with unfractionated heparin or low molecular weight heparin (LMWH) prevents recurrence in these patients (17). However, screening for antiphospholipid antibodies may be appropriate in patients experiencing fetal loss. In addition, there is insufficient evidence of an association, and therefore, insufficient evidence to either screen for or treat women with inherited thrombophilias and obstetric histories that include complications such as IUGR or preeclampsia.
Laboratory Testing for Thrombophilias
Whenever possible, laboratory testing should be performed remote (after 6 weeks) from the thrombotic event and while the patient is not pregnant and not taking anticoagulation or hormonal therapy.
Recommended tests are listed below (18):
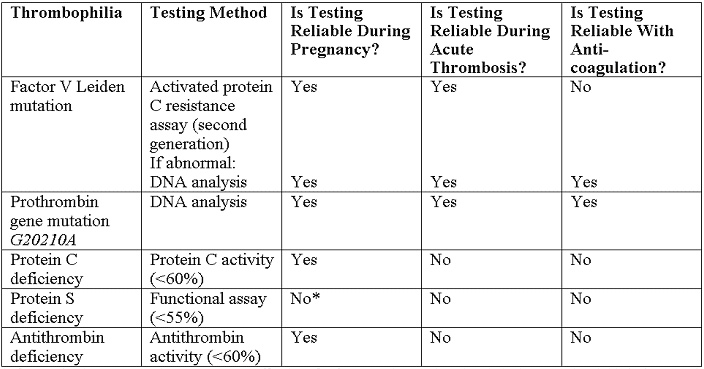
*NOTE - If screening in pregnancy is necessary, cutoff values for free protein S antigen levels in the second and third trimesters have been identified at less than 30% and less than 24%, respectively.
Anticoagulant Regimens for Pregnant Women
Given the risk and benefit ratio of unfractionated heparin, LMWH generally is the preferred agent for prophylaxis in pregnancy. All patients with inherited thrombophilias should undergo individualized risk assessment, which may modify management decisions. The need to adjust LMWH dose according to anti-Xa levels is controversial. Various unfractionated heparin and LMWH regimens are described below (18):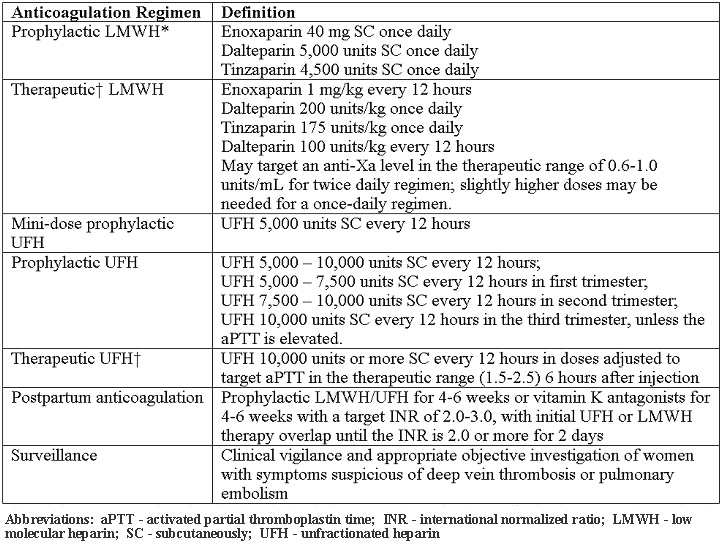
*Although at extremes of body weight, modification of dose may be required
Also referred to as weight adjusted, full treatment dose
Recommended Thromboprophylaxis for Pregnancies Complicated by Inherited Thrombophilias:
The decision to treat with thromboprophylaxis, anticoagulant therapy, or no pharmacologic treatment (antepartum surveillance) is influenced by the venous thromboembolism history, severity of inherited thrombophilia, and additional risk factors. All patients with inherited thrombophilia should undergo individualized risk assessment, which may modify management decisions. The decision regarding intensity of treatment may be shaped by other risk factors, such as cesarean delivery, prolonged immobility, obesity, and family history of thrombophilia or venous thromboembolism. Our recommendations are:
- Low-risk thrombophilia (factor V Leiden heterozygous; prothrombin G20210A heterozygous; protein C or protein S deficiency) patients without previous history of venous thromboembolism (VTE): surveillance without anticoagulation therapy or prophylactic LMWH or UFH is generally recommended. In postpartum period surveillance without anticoagulation therapy or postpartum anticoagulation therapy if the patient has additional coagulation factors (e.g., first-degree relative with history of a thrombotic episode before age 50 years, obesity, prolonged immobility).
- Low-risk thrombophilia patients with a single previous episode of VTE not receiving long-term anticoagulation therapy: prophylactic or intermediate-dose LMWH/UFH or surveillance without anticoagulation therapy is recommended. In postpartum period anticoagulation therapy or intermediate-dose of LMWH/UFH.
- High-risk thrombophilia (antithrombin deficiency, double heterozygous for prothrombin G20210A mutation and factor V Leiden; factor V Leiden homozygous or prothrombin G20210A mutation homozygous) patients without previous VTE: prophylactic LMWH or UFH and in postpartum anticoagulation therapy.
- High-risk thrombophilia patients with a single previous episode of VTE not receiving long-term anticoagulation therapy: prophylactic, intermediate-dose, or adjusted dose LMWH/UFH regimen; and in postpartum period anticoagulation therapy or intermediate or adjusted-dose LMWH/UFH for 6 weeks is usual recommendations. Therapy level should be at least as high as antepartum treatment.
- No thrombophilia with previous single episode of VTE without and associated risk factor (idiopathic) not receiving long-term anticoagulation therapy: prophylactic-dose of LMWH or UFH are recommended in antepartum period and in postpartum period anticoagulation therapy.
- Thrombophilia or no thrombophilia with two or more episodes of VTE receiving long-term anticoagulation: therapeutic-dose of LMWH or UFH during antepartum period is recommended and in postpartum period resumption of long-term anticoagulation therapy.
Intrapartum Management for Thrombophilic Patients
The use of pneumatic compression boots or elastic stockings should be considered for patients with a known thrombophilia until they are ambulatory postpartum. In addition, intrapartum prophylaxis with unfractionated heparin should be considered in patients at higher risk. Regardless of whether the patient is receiving prophylactic, intermediate, or therapeutic doses of LMWH, consideration should be given to substituting a comparable dose of unfractionated heparin at 36 weeks of gestation to permit induction of neuroaxial anesthesia during labor and delivery (19). Alternatively, adjusted-dose subcutaneous LMWH or unfractionated heparin can be discontinued 24-36 hours before an induction of labor or scheduled cesarean delivery to avoid the anticoagulant effect during pregnancy. Patients receiving prophylactic anticoagulation should be instructed to withhold their injections at the onset of labor. If vaginal or cesarean delivery occurs more than 4 hours after a prophylactic dose of unfractionated heparin, the patient is not at significant risk of hemorrhagic complications. Beyond 12 hours after a prophylactic dose or 24 hours after a therapeutic dose of LMWH, spinal anesthesia should not be withheld because the risk of procedure-related bleeding is limited (20). Patients receiving unfractionated heparin or LMWH who require rapid reversal of the anticoagulant effect for delivery can be treated with protamine sulfate (20). In addition, antithrombin concentrates can be used in anti-thrombin-deficient patients in the peripartum period.
Postpartum Management
Postpartum doses of unfractionated heparin or LMWH should be equal to or greater than antepartum therapy. Unfractionated heparin or LMWH can be restarted in 4-6 hours after vaginal delivery or 6-12 hours after cesarean delivery. Patients who will be treated with warfarin may begin therapy immediately after delivery. The initial dose of warfarin should be 5 mg daily for 2 days, with subsequent doses determined by monitoring the international normalized ratio (INR). To avoid paradoxical thrombosis and skin necrosis from the early antiprotein C effect of warfarin, women should be maintained on therapeutic doses of unfractionated heparin or LMWH for 5 days and until the INR is therapeutic (2.0-3.0) for 2 consecutive days. Because warfarin, LMWH, and unfractionated heparin do not accumulate in breast milk and do not induce an anticoagulant effect in the infant, these anticoagulants are compatible with breastfeeding (21), (22).
The risk of VTE among women using estrogen-containing oral contraceptives increases 35-99-fold and 16-fold among women heterozygous for factor V Leiden and prothrombin G20210A mutation, respectively (23). The annual risk of VTE is 5.7 per 10,000 among factor V Leiden carriers, compared with 28.5 per 10,000 among factor V Leiden heterozygous women using estrogen-containing contraceptives, relative risk of 34.7 (24). Therefore, alternative methods, such as intrauterine devices (including those containing progestin), progestin-only pills or implants, and barrier methods, should be considered. However, screening all women for thrombophilias before initiating combination contraception is not recommended.
Fetal Thrombophilia, Perinatal Stroke and Cerebral Palsy
Thrombophilia describes a spectrum of congenital or acquired coagulation disorders associated with venous and arterial thrombosis. These disorders can occur in the mother or in the fetus, or in both concomitantly. Fetal thrombophilia has a reported incidence of 2.4 to 5.1 cases for every 100,000 births (25). Whereas maternal thrombophilia has a subsequently higher incidence, both maternal and fetal thrombophilia can lead to adverse maternal and fetal events. Thrombophilia leads to thrombosis at the maternal or fetal interface. When thrombosis occurs on the maternal side, the consequence may be severe preeclampsia, IUGR, abruption placenta, or fetal loss. Thrombosis on fetal side can be a source of emboli that bypass hepatic and pulmonary circulation and travel to the fetal brain (25). As a result, the newborn can sustain a catastrophic event such as perinatal arterial stroke via arterial thrombosis, cerebral sinus venous thrombosis, or renal vein thrombosis.
Perinatal stroke is defined as a cerebrovascular event that occurs between 28 weeks of gestation and 28 days of postnatal age (26). Incidence is approximately 17 to 93 cases for every 100,000 live births (26). Neonatal stroke occurs in approximately 1 of every 4,000 live births. In addition, 1 in every 2,300 to 4,000 newborns is given a diagnosis of ischemic stroke in the nursery (26). Arterial ischemic stroke in the newborn accounts for 50% to 70% of cases of congenital hemiplegic cerebral palsy. Factor V Leiden mutation, prothrombin gene mutation, and a deficiency of protein C, protein S, and antithrombin III have taken together in two studies, been identified in more than 50% of cerebral ischemic strokes (27). In addition to these thrombophilias, important risk factors for perinatal and neonatal stroke include:
- Thrombosis in placental villi or vessels;
- Infection;
- Use of an intravascular catheter.
What causes perinatal stroke?
The mechanism that underlies perinatal stroke is a thromboembolic event that originates from either an intracranial or extracranial vessel, the heart, or the placenta (27). A recent meta-analysis found a statistically significant correlation between protein C deficiency, MTHFR C677T, and the first occurrence of arterial ischemic stroke in pediatric population (28). The brain is the largest and most vulnerable fetal organ susceptible to thrombi that are formed either in the placenta or elsewhere. The presence of severe fetal vascular lesions correlates highly with neurologic impairment and cerebral palsy. A pathologic finding, fetal thrombotic vasculopathy (FTV), has been associated with brain injury.
Cerebral palsy is the most common chronic motor disability of childhood. Approximately 2 to 2.5 of every 1,000 children are given a diagnosis of this disorder every year (29). The condition appears early in life; it is not the result of recognized progressive disease. Risk factors for cerebral palsy are multiple and heterogenous: prematurity, hypoxia and ischemia, and thrombophilia. Although thrombophilia is a recognized risk factor for cerebral palsy, the strength of the association has still not been fully investigated. Regrettably for patients and their offspring, evidence about the relationship between thrombophilia and an adverse neurologic outcome is insufficiently strong to offer much in the way of definitive recommendations for the obstetricians. We suggest, some tentative recommendations on management: consider screening when cerebral palsy occurs in association with perinatal stroke, fetal and maternal screening for thrombophilia can be performed (27), (30). The recommended thrombophilia panel comprises tests for:
- Factor V Leiden;
- Prothrombin G20210A;
- Anticardiolipin antibody;
- MTHFR mutation.
Family screening has also been suggested in cases of 1) multiple prothrombotic risk factors in an affected newborn, and 2) a positive family history. The cost-effectiveness of screening for thrombophilia has not been evaluated in prospective studies, because the positive predictive value of such screening is extremely low.
Summary
Inherited thrombophilias are a heterogenous group of coagulation disorders that predispose individuals to thromboembolic events. They are major risk factors for thromboembolism during pregnancy and the puerperium. In addition, thrombophilias have been implicated in a variety of adverse obstetric events, including pregnancy loss (especially fetal death), preeclampsia, placental abruption, and IUGR. The pathophysiology is uncertain but is thought to involve thrombosis in the uteroplacental circulation, leading to infarction and placental insufficiency. Hence, anticoagulation therapy has the potential to improve obstetric outcome in women with heritable thrombophilias. Pregnancy outcomes in asymptomatic women with inherited thrombophilias are often good. Therefore, routine treatment with thromboprophylaxis may not be warranted in these women. Inherited thrombophilia testing in women who have experienced recurrent fetal loss or placental abruption is not recommended because it is unclear whether anticoagulation reduces recurrence. Because of lack of association between the MTHFR mutation and negative pregnancy outcomes, screening with fasting homocysteine levels or MTHFR mutation analyses is not recommended. Screening for inherited thrombophilias should include factor V Leiden mutation; prothrombin G20210A mutation; and antithrombin, protein C, and protein S deficiencies. All patients with inherited thrombophilias should undergo individualized risk assessment, which may modify management decisions. Postpartum warfarin, LMWH, and unfractionated heparin anticoagulation may be used in women who breastfeed. A mother whose baby has been given a diagnosis of thrombophilia and fetal or neonatal stroke can be offered thromboprophylaxis (heparin and aspirin) during any subsequent pregnancy. The usefulness of this intervention has not been well studied and is based solely on expert opinion, however, so it is imperative to counsel patients on the risks and benefits of prophylactic therapy beforehand.
Acknowledgement: Gratitude is expressed to Dr. John R. Higgins, Professor of Obstetrics and Gynaecology, Head of College of Medicine and Health, University College Cork, Cork University Maternity Hospital, Wilton, Cork, Ireland for serving as reviewer and helpful suggestions in compiling the manuscript.
References
- Paidas MJ, Ku DH, Lee MJ, et al. Protein Z, protein S levels are lower in patients with thrombophilia and subsequent pregnancy complications. J Thromb Haemost 2005;3:497-501. (Level II-3)
- Chang J, Elam-Evans LD, Berg CJ, et al. Pregnancy-related mortality surveillance United States, 1991-1999. Morb Mortal Wkly Rep Survell Summ 2003;52:1-8. (Level II-3)
- Franco RF, Reitsma PH. Genetic risk factors of venous thrombosis. Hum Genet 2001;109:369-384. (Level III)
- Zotz RB, Gerhardt A, Scharf RE. Inherited thrombophilia and gestational venous thromboembolism. Best Pract Res Clin Haematol 2003;16:243-259. (Level III)
- Gerhardt A, Scharf RE, Beckman MW, et al. Prothrombin and factor V mutations in women with a history of thrombosis during pregnancy and the puerperium. N Engl J Med 2000;342:374-380. (Level II-3)
- De Stefano V, Leone G, Mastranglo S, et al. Thrombosis during pregnancy and surgery in patients with congenital deficiency of antithrombin III, protein C, protein S. Thromb Haemost 1994;71:799-800. (Level III)
- Goodwin AJ, Rosendaal FR, Kottke-Marchant K, et al. A review of the technical, diagnostic, and epidemiologic considerations for protein S assays. Arch Pathol Lab Med 2002;126:1349-1366. (Level III)
- Carraro P. Guidelines for the laboratory investigation of inherited thrombophilias. Recommendations for the first level clinical laboratories. European Communities Confederation of Clinical Chemistry and Laboratory Medicine. Working Group on Guidelines for Investigation of Disease. Clin Chem Lab Med 2003;41:382-391. (Level III)
- Domagala TB, Adamek L, Nizankowska E, et al. Mutations C677T and A1298C of the 5,10-methylenetetrahydofolate reductase gene and fasting plasma homocysteine levels are not associated with the increased risk of venous thromboembolic disease. Blood Coagul Fibronolysis 2002;13:423-431. (Level II-3)
- de Heijer M, Willems HP, Blom HJ, et al. Homocysteine lowering by B vitamins and the secondary prevention of deep vein thrombosis and pulmonary embolism: A randomized, placebo-controlled, double-blind trial. Blood 2007;109:139-144. (Level I)
- Facchinetti F, Marozio L, Frusca T, et al. Maternal thrombophilia and the risk of recurrence of preeclampsia. Am J Obstet Gynecol 2009;200:46.e1-46.e5
- Lissalde-Lavigne G, Fabbro-Peray P, Cochery-Nouvellon E, et al. Factor V Leiden and prothrombin G20210A polymorphisms as risk factors for miscarriage during a first intended pregnancy: the matched case-control 'NOHA first' study. J Thromb Heamost 2005;3:2178-2184. (Level II-2)
- Silver RM, Zhao Y, Song Y, et al. Prothrombin gene G20210A mutation and obstetric complications. Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units (NICHD MFMU) Network. Obstet Gynecol 2010;115:14-20. (Level II)
- Infante-Rivad C, Rivard GE, Yotov WV, et al. Absence of association of thrombophilia polymorphisms with intrauterine growth restriction. N Enlg J Med 2002;347:19-25. (Level II-2)
- Nurk E, Tell GS, Refsum H, Ueland PM, et al. Factor V Leiden, pregnancy complications and adverse outcomes: the Hordaland Homocysteine Study. QJM 2006;99:289-298. (Level II-2)
- Brill-Edwards P, Ginsberg JS, Gent M, et al. Recurrence of clot in This Pregnancy Study Group. Safety of withholding heparin in pregnant women with a history of venous thromboembolism. Recurrence of Clot in This Pregnancy Study Group. N Engl J Med 2000;343:1439-1444. (Level I)
- Kanndorp D, Di Nisio M, Goddijn M, et al. Aspirin or anticoagulants for treating recurrent miscarriage in women without antiphospholipid syndrome. Cochrane Database of Systematic Reviews 2009; Issue 1. Art. No.: CD004734. DOI: 10.1002/14651858.CD004734.pub3. (Level III)
- American College of Obstetricians and Gynecologists. Inherited thrombophilias in pregnancy; practice bulletin No. 124, September 2011. Obstet Gynecol 2011;118:730-740
- Horlocker TT, Wedel DJ, Benzon H, et al. Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuroaxial Anesthesia and Anticoagulation). Reg Anesth Pain Med 2003;28:172-197. (Level III)
- Duhl AJ, Paidas MJ, Ural SH, et al. Antithrombotic therapy and pregnancy: consensus report and recommendations for prevention and treatment of venous thromboembolism and adverse pregnancy outcomes. Pregnancy and Thrombosis Working Group. Am J Obstet Gynecol 2007;197:457.e1-457.e21
- Richter C, Sitzmann J, Lang P, et al. Excretion of low molecular weight heparin in human milk. Br J Clin Pharmacol 2001;52:708-710. (Level III)
- Clark SL, Porter TF, West FG. Coumarin derivatives and breastfeeding. Obstet Gynecol 2000;95:938-940
- Gomes MP, Deitcher SR. Risk of venous thromboembolic disease associated with hormonal contraceptives and hormone replacement therapy: a clinical review. Arch Intern Med 2004;164:1965-1976. (Level III)
- American College of Obstetricians and Gynecologists. Use of hormonal contraception in women with coexisting medical conditions. ACOG Practice Bulletin No. 73. Obstet Gynecol 2006;107:1453-1472. (Level III)
- Lee J, Croen LA, Backstrand KH, et al. Maternal and infant characteristics associated with perinatal arterial stroke in the infant. JAMA 2005;293:723-729
- Nelson KB. Thrombophilias, Thrombosis and Outcome in Pregnancy, Mother, and Child Symposium. Society of Maternal-Fetal Medicine 26th Annual Meeting. Miami Beach, FL; 2006
- Nelson KB, Lynch JK. Stroke in newborn infants. Lancet Neurol 2004;3:150-158
- Winter S, Autry A, Boyle C, et al. Trends in the prevalence of cerebral palsy in a population-based study. Pediatrics 2002;110:1220-1225
- American College of Obstetricians and Gynecologists and American Academy of Pediatrics. Neonatal Encephalopathy and Cerebral Palsy: Defining the Pathogenesis and Pathophysiology. Washington DC: The American College of Obstetricians and Gynecologists; September 2003
- Gibson CS, MacLennan AH, Goldwater PN, et al. Antenatal causes of cerebral palsy: associations between inherited thrombophilias, viral and bacterial infection, and inherited susceptibility to infection. Obstet Gynecol Surv 2003;58:209-220
Published: 4 June 2012
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


