Doppler Ultrasonography in Obstetrics
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC).
The advent of sonography has changed the practice of obstetrics by providing a window to the womb through which the anatomic structure of the fetus can be evaluated. The addition of Doppler flow studies of maternal and fetal vessels has provided a tool where the physiology of the maternal-fetal unit can be assessed. This information can provide the physician and the patient with vital information for a subsequent approach to the pregnancy. The utilization of real-time scanning and the improvement in image resolution following the introduction of high frequency probes have enabled accurate diagnosis and revolutionized the diagnostic as well as therapeutic management of different clinical conditions. Although Satomura demonstrated as early as 1957 that the Doppler technique had the capacity to evaluate blood flow, nearly two decades passed before this technique could be utilized in the assessment of blood flow in fetal umbilical arteries. Doppler flow studies of the umbilical artery can reflect abnormalities in "down-stream" or the fetal side of placental resistance, and the assessment of the maternal vasculature evaluates "up-stream" blood flow or the maternal side of placental resistance. Twenty years ago, an evaluation of the pelvic blood circulation would have required angiography.
The purpose of this document is to describe overview of Doppler ultrasonography and clinical utility of Doppler flow studies in the prediction of adverse pregnancy outcomes in low- and high-risk populations. The use of fetal Doppler blood flow studies has become common in the evaluation and management of pregnancies complicated by conditions such as suspected fetal growth restriction and red blood cell isoimmunization to guide intrauterine therapy and delivery. The most commonly assessed Doppler flow studies of the fetus are the umbilical artery and middle cerebral artery (MCA). Doppler flow studies of MCA are used in the assessment of the fetus at risk of anemia and growth restricted fetus.
Principles and Methods of Doppler Ultrasonography Examination
Doppler Methods
The Doppler phenomenon was first described by Christian Doppler in the 19th century. In its simplified version, this phenomenon is experienced in daily life by all of us. The pitch of the sound of a moving object (e.g. train) is altered when the distance between the observer and the source of sound changes. This change caused by a relative motion between the observer and the object is known as the Doppler shift and is a consequence of the Doppler phenomenon. Accordingly, when the frequency of sound emitted from a stationary source is fixed, and its insonation angle is known, the Doppler shift (i.e. the difference between the emitted and the reflecting frequency) can be calculated, as it is correlated to the velocity of the relative movement between the target and the transducer. This relation is defined by the formula: f D = 2f0v cos θ/c; where f D is the Doppler shift, f0 is the frequency of the transmitted ultrasound, v is the velocity of sound within the tissue (1).
Continuous Wave (CW) Doppler Ultrasonography
In continuous wave (CW) Doppler, the system features separate emitting and receiving transducers that are arranged in a manner that their insonation axes intersect at a certain range determined separately for each pair of transducers. These transducers operate incessantly and are therefore not limited by a maximum detectable velocity range. All Doppler signals (i.e., many different vessels) originating from a certain range of operating area are superimposed, thus making visualization of vessels impossible (1)(2).
Pulsed Wave (PV) Doppler Ultrasonography
This method takes advantage of range discrimination. In pulsed wave (PV) Doppler, the ultrasonic wave is emitted in a pulsatile fashion. Between the pulses of emission, the same transducer operates as a receiver for the back-scattered echoes. Because the velocity of sound is known and presumed to be constant, it is possible to analyze the back-scattered echo alone from a particular range. A circuit selectively permits only those signals that arrive to the receiver at a given time after the transmission. This allows a precise determination of the size of the sample volume that can be located in a particular area (3). This transmit-and-receive sequence is repeated periodically. The rate at which this is accomplished determines the performance of the pulsed Doppler system. One shortcoming of pulsed Doppler arises from the fact that a new pulse cannot be emitted before the last echo of the preceding pulse has arrived at the transducer. This imposes a limit on the detectable absolute maximum velocity of blood flow, a feature that does not appear with CW Doppler. The maximum Doppler shift frequency that could be measured is related to half the pulse repetition frequency (Nyquist limit). Beyond this limit, Doppler signals will be distorted (aliasing). Aliasing can be corrected by either increasing the pulse repetition frequency or lowering the baseline. Aliasing may occur also in color Doppler imaging. The integration of real-time ultrasonography and pulsed Doppler techniques is referred to as duplex Doppler scanning. The concept of range discrimination described above would not be any benefit without proper guidance to the target organs in the operating area of the pulsed Doppler sample volume. B-mode real-time imaging has provided such guidance. In duplex scanning, it is customary to locate the target with real-time imaging and then to switch the apparatus to the Doppler mode. New electronic probes enable real-time duplex scanning. In this combined mode, however, the velocity range limitation of pulsed Doppler is emphasized to a greater degree.
Color Flow Imaging
Color Doppler Imaging (CDI): The earlier, two-dimensional flow imaging systems were based on CW Doppler and non-real-time scanning (1). In the early 1980s, a real-time, two-dimensional, flow imaging technique that utilized and autocorrelation, processor for the detection of a moving target was introduced. In the current and more sophisticated color Doppler imaging (CDI), color-coded pulsed Doppler information is superimposed on a B-mode ultrasonic image. In this method, color is assigned to flow direction. Customarily, flow toward the Doppler transducer is displayed in red, and flow away from it is shown in blue. The structures that do not move are presented in basic gray-scale image. The color saturation is related to the magnitude of the frequency shift (3). Color flow imaging facilitates the detection of small vessels and slow blood-flow velocity. Color flow imaging is subject to the same limitations as pulsed Doppler.
Color Doppler Energy (CDE): It detects the energy of Doppler signals generated from moving blood. The basic difference between the CDE and conventional CDI can be summarized as follows: (i) Because CDE is not dependent on the mean velocity but on the amplitude, it is angle-dependent; (ii) CDE is able to display lower volumes and velocities; and (iii) CDE is not affected by aliasing. Taken together, all these advantages of CDE over CDI enable the investigation of blood flow with very low velocity, even in the vessels running at 90 degrees to the insonation angle of the ultrasound beam (3). The conventional semiquantitative analysis based on the use of impedance indices is not applicable to CDE studies because CDE does not rely on mean velocity of the blood flow.
Doppler Velocimetry
Qualitative Assessment: In the conventional Doppler equation, the movement of a single target generates a Doppler shift of only one frequency. This condition cannot be obtained in a blood vessel where blood flow comprises numerous blood cells moving at a wide range of different velocities. A Doppler signal detected by the receiving transducer is therefore a combination of different Doppler shift frequencies. This mixture of frequencies is analyzed by an on-line computer and transformed into a series of spectra (Doppler spectrum) that are graphically displayed in real time. In other words, these graphical drawings (flow velocity waveforms) show the relative power of each frequency component that constitutes the entire Doppler signal (1). The mean flow velocity waveform (FVW) is therefore related to three variables: time, frequency, and power. The power of different frequencies relates to volumes of blood moving at that certain velocity. The simplest qualitative method used in interpreting the Doppler data is to decide whether flow is present or not. This can be achieved either visually or by listening to Doppler signals.
Quantitative Assessment: The measurement of the velocity, acceleration, and volume of blood flow can be achieved with Doppler data. When the angle between the ultrasound beam and the longitudinal axis of the vessel is known, the Doppler frequency shift can be changed into velocity. The measurement of this angle is of major importance when calculating velocity. The larger the insonation angle, the greater the chance of error (4)(5). The uterine artery is rather small and has a curved course. The maximum velocity at a given time reflects the fastest frequency obtained from somewhere within the sample volume. Although this calculation is also related to the insonation angle, recent research has demonstrated that the maximum velocity measurements can be applied to Doppler studies. The velocity measurements most commonly used in pulsed Doppler studies are the maximum peak systolic velocity, the highest time-average maximum velocity, and the minimum diastolic velocity (5).
Semiquantitative Assessment: Fundamental ambiguities inherent in Doppler velocity measurements have led to semiquantitative assessment of the FVWs. Here, the relationship between the systolic and diastolic components of the waveform is evaluated, and angle dependence, which is important in quantitative measurements, becomes less important. Different equations have been proposed to define the properties of the Doppler spectrum, the most common in obstetric applications being the pulsatility index (PI) and the resistance index (RI), (also Pourcelot index): PI=S-D/A; RI=S-D/S; S/D ratio -- where S is the maximum peak systolic frequency, D is the end-diastolic, and A is the mean Doppler shift frequency during a cardiac cycle (6). The relation between these indices has been assessed and was found to be highly correlated. PI and RI directly reflect the downstream flow impedance (1)(5)(6). From a theoretical point of view, resistance in the peripheral vessels is defined by the ratio of mean pressure to mean flow. In clinical applications, blood flow is assessed in a pulsatile arterial circulation. Thus, the pressure in this system changes throughout the cardiac cycle. Therefore, peripheral resistance is replaced by vascular impedance (6). In the current literature, both terms are often used interchangeably.
Definitions:
Resistance index (RI): Maximum -- minimum velocity/maximum velocity;
Pulsatility index (PI): Maximum -- minimum velocity/mean velocity.
Safety of Doppler Ultrasonography
Diagnostic ultrasonography has been used in medicine for many decades. To date, at the intensities allowed by regulations, there has been no solid evidence of any detrimental biologic effects in humans. Literally millions of people around the world are exposed each year to medical ultrasonography, a technique that continues to boast an excellent safety record. In USA, more than three scans per live birth are performed annually. Despite proven safety of the method, ultrasonography does not have some inherent bioeffects that should not be ignored and are therefore addressed below. An important factor that plays a major role in the development of biologic effects is the acoustic power of the ultrasonography device, i.e., the amount of energy produced in a unit of time. The duration of the ultrasound pulse, the pulse repetition frequency (the reciprocal of the interval between the pulses), and the duty factor (the pulse duration divided by the pulse repetition frequency) are the most important determinants of acoustic power output. In particular, pulsed Doppler ultrasonography has higher duty factors than do conventional imaging facilities. There are two potential mechanisms through which ultrasonography can produce biologic effects: thermal and mechanical (7). Tissue heating, i.e., the thermal effect, is a consequence of the tissue absorption of the ultrasound wave. Mechanical effects consist of cavitation and radiation forces. The acoustic power, intensity, and related potential of bioeffects depend on the mode of ultrasonography. With Doppler, a longer pulse duration and a generally higher pulse repetition frequency are used, resulting in a higher duty factor, and therefore, in a greater potential for heating. The least energy is produced by real-time imaging and the most with Doppler mode. Color flow imaging falls between these two.
According to the clinical safety statement of the European Federation for Societies of Ultrasound in Medicine and Biology (6)(7), the investigator should bear in mind that pulsed Doppler examination at maximum equipment output power and color flow imaging very small color box may have, in theory at least, some potential biologic effects. There are genuine concerns about safety of color and pulsed Doppler examinations in the first trimester, during the period of fetal organogenesis, in view of the closeness of the transvaginal transducer to the fetus and the higher intensities generated, especially by pulsed Doppler examination. Some leading practitioners believe that in first trimester, Doppler may have the capacity to thermally disturb embryologic development (6). The main danger would appear to be that of heating; it could be argued that, as the fetal skeleton develops after the critical period of organogenesis, the main contribution to heating (i.e., bone) does not come into equation. Nevertheless, there is a body of opinion that believes that first trimester Doppler should be performed only for women requesting termination of pregnancy. However, if the aim of the study was to determine whether first trimester fetal circulatory parameters were predictive of pathology developing later in pregnancy, confining such studies to pregnancies planned for termination would be of no value. In an editorial (8) in Ultrasound in Obstetrics and Gynecology, the journal editors decided that papers on first trimester Doppler should be accepted for publication providing certain safeguards were in place. All equipment used should display the safety limits of thermal index (TI) and the mechanical index (MI), and these must be below the current Food and Drug Administration (FDA) limits of safety. Exposure time have to be based on the as-low-as-reasonably-
In summary, the investigator is particularly advised to lower actively the output intensity when scanning in early pregnancy (TI<1) and not to increase the pulse repetition frequency. With advancing gestation, the examination time should be kept short if TI>1 (safe exposure time <2.6 minutes at 39ºC [T=2] and only 38 seconds at 40ºC [T=3]). Also, in order to prevent the non-thermal effects, sound pressure should be <1MPa and MI<1. Nevertheless, particular care should be taken to minimize the exposure times and the output power of the scanners in order not to have the enviable record of ultrasonography for safety be challenged (7). This becomes even more important now and in future if the trend for the manufacturers to increase the output levels of the new equipment continues. There are times in which CDI may allow one to answer a clinical question concerning fetal viability quickly and allow a shorter total examination to be performed. For example, in the presence of poor image quality because of maternal adipose tissue, CDI will facilitate the documentation during the first trimester, is unnecessary for documentation of embryonic or fetal life when conventional B-mode real-time scanning clearly demonstrates cardiac or fetal motion.
Clinical Applications of Doppler Ultrasonography in Obstetrics
Fetal Doppler Ultrasonography Examinations
The fetal responses and adaptation to changes in the intrauterine environment can be investigated by looking at the fetal blood circulation. Both arterial and venous fetal circulation has been widely documented. Initial reports documented Doppler velocimetry of the umbilical arteries (UAs), the umbilical venous flow and the fetal descending aorta.
Arterial
Middle Cerebral Artery: The cerebral blood flow in the human fetus has been demonstrated by Doppler imaging of the internal carotid, middle cerebral, posterior cerebral, and anterior cerebral arteries (9). With accumulating experience, the middle cerebral artery (MCA) has become an important part of the fetal Doppler assessments because it is easier to detect and measure cerebral vessels and has a high sensitivity in the detection of fetal intrauterine growth restriction (IUGR) and related complications (10). The MCA can be visualized in a transverse axial view of the fetal head at a slightly more caudal plane than the one used for biparietal diameter measurements. At this level, which includes the cerebral peduncles, the MCA can be seen as major lateral branches of the circle of Willis coursing anterolaterally towards the lateral edge of the orbits at the level of the lesser wing of the sphenoid bone into the lateral sulcus and disappearing onto the cerebral fossa between the insula and the temporal lobe. The sample volume should be placed at the proximal part of MCA. During pregnancy, there is a continuous forward flow in all cerebral arteries throughout the cardiac cycle.
n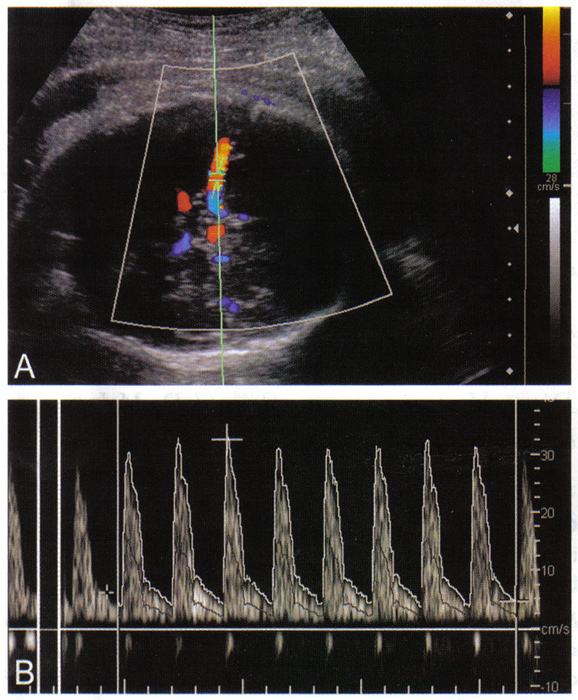
Fig. 1. A. Color Doppler mapping of the Circle of Willis, with cursor placement in the proximal middle cerebral artery and angle correction. B. Doppler spectral waveform of the middle cerebral artery in a fetus of 30 weeks of gestation, measuring peak systole, time average maximum velocity, and end diastole.
Thoracic Aorta: By attaching the real-time B-mode ultrasonography and pulsed Doppler transducers to each other at a fixed angle, volume-flow measurements in the descending aorta can be performed. The blood flow in the descending fetal aorta is approximately 185 to 261 mL/min-1kg-1 (11). The weight-related blood flow remains stable until 37 weeks, after which it falls slightly. In the aorta of normal fetuses there is continuous forward flow throughout the whole cardiac cycle, which is caused the low vascular resistance in placental circulation. The PI characterizes the aortic vascular impedance, which is stable during the last trimester of gestation. The Doppler velocimetry measurements of the thoracic aorta are performed in that portion of the descending aorta above the diaphragm. In growth-restricted fetuses and in fetuses in distress, characteristic changes of the aortic FVW have been reported. As a consequence of hypoxia, the vascular impedance increases, indicated by the absence of the end-diastolic velocity (11). In this study (12) the purpose was to evaluate the association between an abnormal aortic isthmus blood flow index and postnatal neurodevelopmental outcome in fetuses with placental circulatory insufficiency. An isthmus blood flow index cut-off value of 0.70 was associated with the highest overall positive and negative predictive values. The pulsatility index in the umbilical artery did not provide any significant contribution in the explanation of the outcome. The isthmic blood flow index can help to identify a subgroup of fetuses with placental circulatory insufficiency that might benefit from early delivery.
n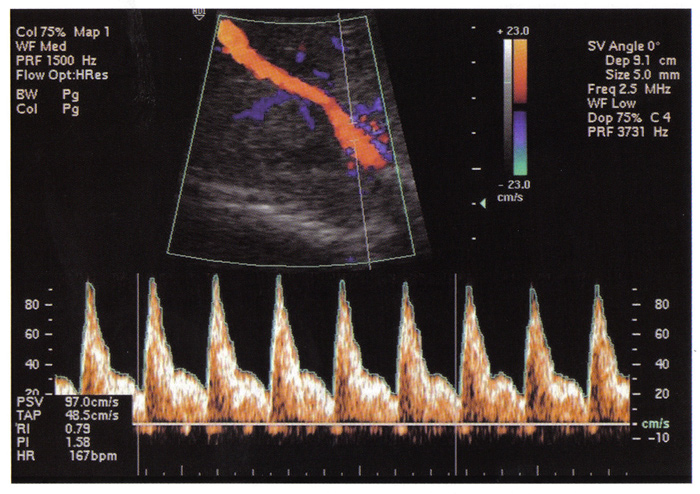
Fig. 2. The visualization of thoracic aorta and its flow velocity waveform. Pulsatility index: 1.58 indicates normal blood flow
Renal, Splenic, Adrenal, and Hepatic Arteries: With recent advances of Doppler ultrasonography equipment, it has become feasible to study small fetal vessels such as renal, splenic, mesenteric, adrenal and hepatic arteries. Doppler velocimetry of the renal arteries can be performed in a longitudinal or coronal plane of section that identifies the fetal kidney and the renal artery as they arise from descending aorta. In small for gestational age (SGA) fetuses the renal vascular impedance is higher than in normal fetuses (13). The difference becomes more evident in the presence of oligohydramnios. The fetal splenic artery can be visualized posterior to the stomach as it arises from the celiac axis and courses to the hilum of the spleen in a transverse plane of section of the fetal abdomen at the level of the fetal stomach. Different groups have explored the utility of splenic artery velocimetry in the prediction of fetal hypoxia. The literature provides little information on the fetal adrenal blood flow. It is possible to visualize the adrenal vessels in 84% of the cases. The adrenal artery PI decreases with advancing gestation. Few investigators attempted to measure the blood flow in hepatic arteries. Fetal hepatic artery FVW could be regarded as being subject to redistribution as are the cerebral, adrenal, and coronary arteries.
Atrioventricular (AV) Inflow: It is assessed at the level of mitral and tricuspid valves from the apical four-chamber view. FVW across the atrioventricular (AV) valves displays a biphasic forward flow consisting of early (E wave) and late (A wave) inflow velocity peaks, reflecting early ventricular filling during ventricular diastole and active ventricular filling during atrial systole, respectively. The inflow across the AV valves depends on the ventricular compliance and preload. No download stream impedance as expressed by descending aorta and UAs vascular resistance (14). The earlier monophasic AV inflow becomes biphasic as early as 8 weeks. Throughout gestation, the inflow during the atrial contraction is higher than that during the early ventricular filling. Both the E and A inflow velocities increase with advancing gestation and almost equalize at term. Consequently, the E/A ratio increases from 0.5 in the late first trimester to 0.8 to 0.9 in late pregnancy (15). Throughout gestation, the transtricuspid time-averaged velocity (TAV) is higher than transmitral TAV. These findings indicate the right ventricular predominance and its lower compliance when compared with the left ventricle. After birth, during the first year of life, inflow velocities remain unchanged, and the ventricular filling shifts from right-sided dominance to a left-sided dominance. In small-for-gestational age (SGA) fetuses (<5th percentile), both transmitral and transtricuspid flow velocities decrease because of reduced volume flow (16). This seems to be determined by reduced preload as a consequence of reduction in central venous return.
Branch Pulmonary Arteries and Ductus Arteriosus (DA): Total pulmonary blood flow accounts for 13% of the combined cardiac output at 20 weeks and for 25% at 30 weeks (17). This change is attributable to a 1.5-fold decrease in pulmonary vascular resistance. After 30 weeks of gestation the proportion of pulmonary blood flow with regards to the combined cardiac output remains unchanged (17). The success rate of obtaining Doppler measurements from the proximal part of the pulmonary arteries varied between 85% and 98%. As well, the distal branches of the pulmonary arteries can be visualized. The FVW obtained from the proximal part of the branch pulmonary artery is characterized by an initial rapid acceleration of flow, followed by a subsequent similar rapid deceleration, producing a systolic peak with a needle-like appearance. The systolic peak is followed by an early diastolic reversed flow and a reduced or absent end-diastolic flow. Taken together, these indicate a high-resistance low-volume circuit in the fetal pulmonary circulation. During the second half of pregnancy, the peak systolic velocity and time-to-peak-velocity interval in branch pulmonary arteries increase with decreasing vascular impedance. This decrease in vascular impedance determined by a low PI continues up to 34 weeks in the proximal part and up to 31 weeks in the distal parts of the branch pulmonary arteries (18). During the end of third trimester, the pulmonary resistance increases because of the acquired vasoconstriction, the consequence of which is the diversion of the combined cardiac output to the systemic circulation. Recently, the clinical value to pulmonary artery Doppler velocimetry in the diagnosis of pulmonary hypoplasia and fetal asphyxia in intrauterine growth restricted (IUGR) fetuses has been assessed (18). In fetuses with pulmonary hypoplasia, the vascular impedance in the pulmonary artery was higher and peak systolic velocity was lower than in healthy fetuses (18). As well, in IUGR fetuses the PI was higher than in normal fetuses. Moreover, in the group of hypoxic fetuses diagnosed by cordocentesis, there was a significant relationship between the severity of the hypoxia and the PI values.
The DA connects the pulmonary trunk to the descending aorta during fetal life, acting as a right-to-left shunt. The construction of DA in utero may lead to fetal pulmonary hypertension. The ductal blood flow can be assessed by fetal echocardiography, and ductal constriction can be readily diagnosed. The success rate of obtaining adequate signals is as high as 94%. The peak systolic velocity in the human DA varies from 50 to 200 cm/sec and diastolic velocity varies from 6 to 30 cm/sec (19). Both velocities increase with advancing gestation, where as the PI remains unchanged. The FVW of the DA displays a continuous forward flow throughout the cardiac cycle. Indomethacin has been widely used to prevent preterm labor before 34 weeks. This drug has a reversible constrictive effect on the DA (19). In addition, 40% to 60% of the treated fetuses develop tricuspid regurgitation. The ductal constriction is considered as being severe if the PI value is less than 1. During fetal ductal occlusion, no blood flow could be detected in DA using CDI or pulsed Doppler ultrasonography.
Placental Doppler Ultrasonography Examinations
Umbilical Arteries (UAs)
Umbilical arteries (UAs) and uterine Doppler velocimetry studies enable the assessment of the uteroplacental and fetoplacental units. The 40% of the combined fetal ventricular output is directed to the placenta by two umbilical arteries (UAs). The Doppler velocimetry measurements of umbilical blood flow have become feasible by the introduction of real-time pulsed Doppler ultrasonography. The volume in the UAs increases with advancing gestation. Concomitantly, the high vascular impedance detected in first trimester gradually decreases. This decrease in vascular impedance has been attributed to growth of the placental unit and increase in the number of the functioning vascular channels (1)(2). Low vascular impedance allows a continuous forward blood flow in the UA FVW throughout the cardiac cycle. Earlier research has shown that the Doppler UA FVW studies predict the most compromised fetuses in high-risk pregnancy. UA velocimetry assessment decreases the perinatal mortality from IUGR without any increase in rate of unnecessary obstetric interventions in high-risk pregnancies (20). The current data, however, question the benefits of using UA Doppler velocimetry as a routine screening test for fetal hypoxia or acidosis in low-risk pregnancies.
n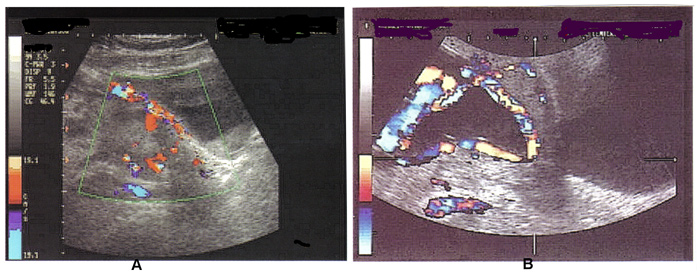
Fig. 3 A. Color Doppler ultrasonography of placenta accreta; B. Color Doppler ultrasonography of vasa previa
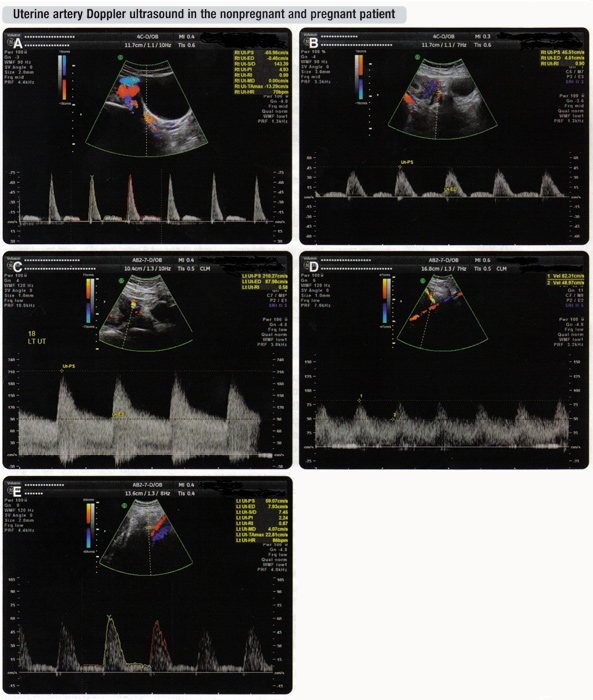
Uterine Arteries
In normal pregnancy, placental trophoblast cells invade the inner third of myometrium and migrate the entire length of the maternal spiral arteries. Remodeling of these high resistance arteries results in a low resistance and high flow state in the intervillous space, which optimizes delivery of oxygen and nutrients to the fetus. This change in resistance is reflected in uterine artery Doppler studies by a high diastolic velocity with continuous flow during diastole. In women who develop preeclampsia there is failure of trophoblast invasion of the uterine vasculature with the result that the spiral arteries retain the muscle elastic coating and impedance to blood flow persists (21). A similar mechanism of failed trophoblast invasion and high resistance has been described in women who subsequently deliver infants with growth restriction (22). Theoretically, a pathological increase in placental vascular resistance should be detectable by abnormal Doppler flow studies of the maternal uterine vessels, and this could offer the potential to detect women at risk for diseases like preeclampsia and fetal growth restriction.
Criteria for an abnormal test: The majority of research has centered on an elevation in the RI or PI, or the persistence of a uterine artery diastolic notch to detect the presence of increased uteroplacental vascular resistance. Criteria for an abnormal RI have varied from a single cutoff (eg, RI>0.58) to a percentile cutoff value (eg, 75th, 90th, 95th). The FASTER study, a large prospective observation study, found a uterine artery Doppler RI value above the 75th percentile at 10-14 weeks' gestation to predict a 5.5-fold higher likelihood of subsequent growth restriction than those with a lower value (22). It appears that as the impedance to flow increases in the placenta there is momentary closure of the uterine artery in late systole or early diastole, or an increase in downstream resistance as the relatively inflexible distal artery recoils from distention caused by the systolic pulse. This is manifested as an early diastolic notch in the Doppler wave form (shown below). Most studies use subjective criteria for the definition of a diastolic notch, but a drop of at least a 50 cm/s from the maximum diastolic velocity is a reasonable criterion after 20 weeks. In summary, there are no current standards for gestational age at testing or criteria for an abnormal uterine artery Doppler study. Once adequately trained in technique, a reasonable approach would be to use an ultrasound machine with the capability to perform continuous wave and/or pulsed wave Doppler studies of the uterine, arcuate, and sub-placental arteries.
nThere are limited studies of uterine artery Doppler in women with multiple gestations. Until further information is available, the role of uterine artery Doppler screening in women with multiple gestations is unclear, and a negative test should not be used to reassure the patient regarding potential adverse pregnancy outcomes (23). It appears that uterine artery Doppler studies are not helpful in differentiating aneuploidy from euploid fetuses (23). Abnormal uterine artery Doppler studies in the first and second trimester have been associated with subsequent adverse pregnancy outcomes including preeclampsia, fetal growth restriction, and perinatal mortality. However, the predictive value of Doppler testing in low-risk population of women appears to be low, and currently there are no available interventions to prevent adverse outcomes based on an abnormal result (24). Uterine artery Doppler screening of high-risk women (eg, history of chronic hypertension or preeclampsia, prior fetal growth restriction, or stillbirth) with singleton gestations appears to identify those at substantially increased risk for adverse pregnancy outcomes. Abnormal testing in these women could potentially lead to increased surveillance (eg, earlier and more frequent assessment of fetal growth and maternal clinical condition) and interventions that might improve clinical outcomes.
n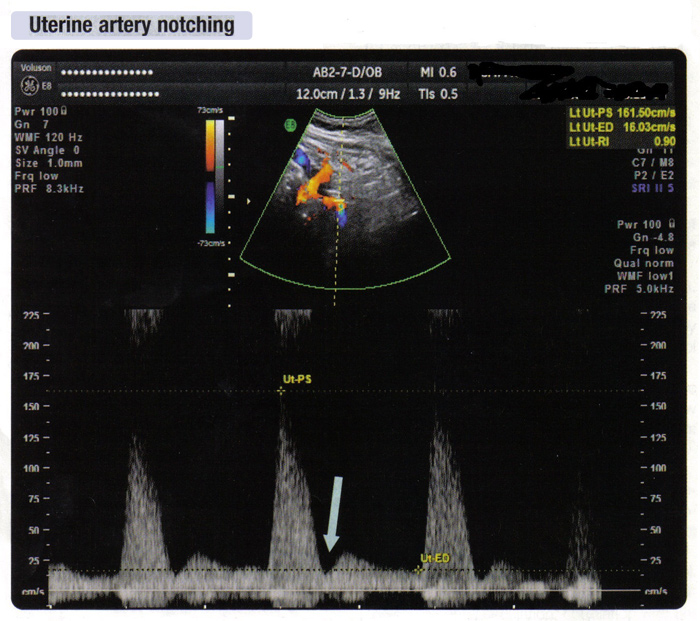
Fig. 5 Uterine artery notching
Doppler Analysis to Assess Effects of Magnesium Sulfate
Magnesium sulfate has become one of the most commonly used drugs in American obstetrics. Indications for use have included management of eclampsia, tocolytic treatment of preterm labor, and most recently, prevention of brain damage in very low birth weight (less than 1,500 g) neonates. The reported cerebral protective effects include prevention of cerebral hemorrhage in the neonatal period as well as cerebral palsy during infancy. Hypothesized pathways of brain injury in very low birth weight neonates include precluding hypoxic-ischemic injury with reperfusion intracerebral bleeding in the first 2-3 days of life and fluctuations in cerebral blood flow associated with neonatal interventions such as mechanical ventilation. The protection against brain injury has been hypothesized to be due to blockade of N-methyl-D-aspartate receptors, by the attenuation of vasoactivity by blockade of calcium channels, and lastly by direct arterial dilation by the magnesium ion (25). In a recent randomized controlled trial to measure Doppler flow indices in the fetal cerebral circulation to determine whether this methodology could estimate magnesium-sulfate induced changes in the cerebral blood flow (26). The study concluded magnesium sulfate had no significant effects on fetal cerebral blood flow analyzed using Doppler. The only parameter in the fetal cerebral circulation significantly modified by magnesium sulfate was the heart rate. The significance of this heart-rate change, vis-à-vis the neuroprotective effects of magnesium sulfate, is unknown.
Summary
Doppler ultrasonography studies in the prediction of adverse pregnancy outcomes in low and high risk populations have an important place in modern obstetrics. Color Doppler energy (CDE) enables a faster and easier acquisition of Doppler signals. Consequently, a very subtle amount of blood flow can be visualized. CDE displays a third parameter of this Doppler signal: the strength or energy. The physical background for Doppler errors needs to be emphasized before any issue on reproducibility can be considered. Blood flow in vessels differs from in vitro flow models. Blood is approximately four times more viscous than water, and it flows in an elastic system. Therefore, in general, the blood-flow profile does not obey the rules that control the flow conditions. The new FDA policy no longer sets an upper limit for the output intensity of obstetric scanners and thus gives the full responsibility to the user with regards to patient safety. In theory, this tempts manufacturers to develop new equipment using higher intensities in order to improve the diagnostic capability and the marketing of their product. Further studies are needed to determine which high-risk conditions are amenable to Doppler screening, what testing regimen is optimal for a normal or abnormal test in low- and high-risk populations, and what interventions based on these findings will improve pregnancy outcomes. At this time, evidence does not support routine screening with Doppler ultrasonography. Use of umbilical artery Doppler should be individualized, and a plan of management based on the results should be put in place. Because standards of the study technique, gestational age, and criteria for an abnormal test are lacking, uterine artery Doppler studies should not be considered to be a required medical practice in low or high risk populations.
Acknowledgment:
Women's Health and Education Center (WHEC) expresses gratitude to Dr. John P. O'Grady, Professor, Obstetrics and Gynecology, Tufts University School of Medicine, Medical Director Mercy Perinatal Service, for his priceless contribution in preparing the series on Doppler Ultrasonography in Modern Obstetrics.
References:
- Burns PN. The physics of Doppler. In Chervenak FA, Isaacson GC, Campbell S (Eds): Ultrasound in obstetrics and Gynecology. Boston, Little, Brown & Co., 1993
- Dickerson KS, Vernon MS, Newhouse VL et al. Comparison of conventional and transverse Doppler sonograms. J Ultrasound Med 1993;12:497-502
- Tekay A, Campbell S. Doppler ultrasonography in obstetrics. In Callen Ultrasonography in Obstetrics and Gynecology. Publisher: W.B. Saunders; 5th edition, 2008
- Maulik D. Hemodynamic interpretation of the arterial Doppler waveform. Ultrasound Obstetrics and Gynecology 1993;3:219-223
- Acharya G, Wilsgaard T, Berntsen GKR et al. Doppler-derived umbilical artery absolute velocities and their relationship to fetoplacental volume blood flow: a longitudinal study. Ultrasound Obstet Gynecol 2005; 25: 444-453
- Tekay A, Campbell S. Doppler ultrasonography in obstetrics; In Callen Ultrasonography in obstetrics and gynecology; 5th edition, Publisher: Saunders, 2008
- Duck FA. Is it safe to use diagnostic ultrasound during the first trimester? Ultrasound Obstet Gynecol 1999;13:385-392
- Campbell S, Platt L. The publishing of papers on first-trimester Doppler. Ultrasound Obstet Gynecol 1999;14:159-160
- Pearce W. Hypoxic regulation of the fetal cerebral circulation. J Appl Physiol 2006;100:731-738
- Bilardo CM, Wolf H, Stigter RH et al. Relationship between monitoring parameters and perinatal outcome in severe, early intrauterine growth restriction. Ultrasound Obstet Gynecol 2004; 23: 119-125
- Del Río M, Martínez JM, Figueras F et al. Doppler assessment of fetal aortic isthmus blood flow in two different sonographic planes during the second half of gestation. Ultrasound Obstet Gynecol 2005; 26: 170-174
- Fouron JC, Gosselin J, Raboisson MJ et al. The relationship between an aortic isthmus blood flow velocity index and the postnatal neurodevelopmental status of fetuses with placental circulatory insufficiency. Am J Obstet Gynecol 2005; 192: 497-503
- Baschat AA. Fetal responses to placental insufficiency: an update. BJOG 2004; 111: 1031-1041
- Hecher K, Snijders R, Campbell S, Nicolaides K. Fetal venous, intracardiac and arterial blood flow measurements in intrauterine growth retardation: relationship with fetal blood gases. Am J Obstet Gynecol 1995; 173: 10-15
- van Splunder P, Stijnen T, Waldimiroff JW. Fetal atrioventricular, venous and arterial flow velocity waveforms in the small-for-gestational-age fetus. Pediatr Res 1997;42:765-772
- Kiserud T, Chedid G, Rasmussen S. Foramen ovales changes in growth-restricted fetuses. Ultrasound Obstet Gynecol 2004; 24: 141-146
- Mitchell JM, Roberts AB, Lee A. Doppler waveforms from the pulmonary arterial system in normal fetuses and those with pulmonary hypoplasia. Ultrasound Obstet Gynecol 1998;11:167-171
- Yoshimura S, Masuzaki H, Miura K et al. Diagnosis of fetal pulmonary hypoplasia by measurement of blood flow velocity waveforms of pulmonary arteries with Doppler ultrasonography. Am J Obstet Gynecol 1999;180:441-443
- Figueras F, Martínez JM, Puerto B et al. Contraction stress test versus ductus venosus Doppler evaluation for the prediction of adverse perinatal outcome in growth-restricted fetuses with non-reassuring non-stress test. Ultrasound Obstet Gynecol 2003; 21: 250-255
- Schwarze A, Gembruch U, Krapp M et al. Qualitative venous Doppler waveform analysis in preterm intrauterine growth-restricted fetuses with ARED flow in the umbilical artery - correlation with short-term outcome. Ultrasound Obstet Gynecol 2005; 25: 573-579
- Axt-Fliedner R, Schwarze A, Nelles I et al. The value of uterine artery Doppler ultrasound in the prediction of severe complications in a risk population. Arch Gynecol Obstet 2005;271:53-58
- Duggoff L, Lynch AM, Cioffi-Ragan D et al. FASTER Trial Research Consortium. First trimester uterine artery Doppler abnormalities predict subsequent intrauterine growth restriction. Am J Obstet Gynecol 2005;193:1208-1212
- Sebire NJ. Opinion: routine uterine artery Doppler screening in two pregnancies? Ultrasound Obstet Gynecol 2002;20:532-534
- Sciscione AC, Hayes EJ. Uterine artery Doppler flow studies in obstetric practice. 2009;201:121-126
- Pearce W. Hypoxic regulation of the fetal cerebral circulation. J Appl Physiol 2006;100:731-738
- Twicler DM, McIntire DD, Alexander JM et al. Effects of magnesium sulfate on preterm fetal cerebral blood flow using Doppler analysis. Obstet Gynecol 2010;115:21-25
Published: 26 January 2011
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com