Recurrent Pregnancy Loss
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC).
Recurrent pregnancy loss (RPL) is a frustrating problem for both patients and physicians. Spontaneous abortion or miscarriage is defined as pregnancy loss before 20 weeks' gestation or fetal weight of 500 g or less. Pregnancy is a complicated process involving many intricate interactions between the fetus and the maternal environment. Pregnancy loss can result from any number of genetic, anatomic, endocrine, immune, or thrombotic disorders, as well as from unknown causes (1). Despite a comprehensive evaluation, the cause of recurrent pregnancy loss (RPL) cannot be identified for more than half of affected couples. Cautious reassurance and encouragement are important -- as is careful monitoring. Recurrent pregnancy losses (RPLs) do not imply inevitability. Even when patients have RPLs without a known etiology, future outcomes may not be dismal. In considering the likelihood of pregnancy loss without regard to specific diagnosis, a woman who has had no losses with pregnancy has a 15% chance of loss with the next pregnancy. For a woman with one unexplained prior loss, the risk of loss with the next pregnancy rises to 13%; and to 30% with two unexplained losses. Even with patients who have had four prior pregnancy losses, however, the chance of carrying the next pregnancy to term is 60% (2). The most important aspects of managing the patient with RPL are frequent communication, education, and emotional support. Cautious reassurance should be offered as the large majority and about 70% of patients will ultimately achieve a successful pregnancy. Careful monitoring is warranted because women with RPL are also at increased risk for other complications of pregnancy such as preterm birth and ectopic pregnancy.
The purpose of this document is to outline the causes of recurrent pregnancy loss (RPL) and their potential therapies, where applicable. Human reproduction is an inefficient process. Only 15% to 30% of fertilized oocytes result in viable pregnancy. After complete evaluation and successful treatment, couples who have experienced spontaneous abortion are often afraid to become pregnant again. These patients need intensive physician support during their first trimester. This review can serve as a useful resource when counseling patients regarding treatment options.
Definition
Recurrent pregnancy loss (RPL) is currently defined as three or more consecutive pregnancy losses of clinically recognized pregnancies prior to the 20th week of gestation (excludes -- biochemical, ectopic and molar pregnancies). Approximately 2% of women have two consecutive pregnancy losses. Traditionally, RPL, which affects approximately 1% of couples, refers to the loss of three or more consecutive pregnancies. However, the risk of third loss after two miscarriages is approximately 30%, whereas the risk after three losses is about 33% (3). Because this difference is very small, many clinicians will begin the evaluation of RPL after two losses. This approach may be especially useful in older women. Primary RPL refers to patients who have never achieved pregnancy, whereas secondary RPL refers to those in whom the miscarriages follow at least one normal pregnancy. The risk of miscarriage is lower in women who carry at least one pregnancy to term, whereas a history of miscarriage increases the chance of miscarriage in subsequent pregnancies. Maternal age is also a strong predictor of recurrent losses.
Epidemiology
Of clinically recognized pregnancies, 10% to 15% result in spontaneous abortion. Most of these pregnancy losses are clinically evident by 12 weeks' gestation. Embryonic or pre-embryonic demise usually occurs at least 1 week prior to the clinical features of pregnancy loss being recognized. The risk of RPL was once thought to increase progressively with each spontaneous abortion and result in a woman having an 80% chance of a subsequent spontaneous abortion after three spontaneous abortions. Currently, it is believed that the spontaneous risk increases with the number of pregnancy losses, but very gradually, and rarely exceeds 40% to 50%. For a woman who has had a live birth, even if she has had multiple spontaneous abortions, her risk of spontaneous abortion in future pregnancies is 30% per pregnancy (4).
Etiology and Risk Factors
The following causes of RPL are widely accepted. The workup of RPL is negative in more than 50% of patients (5). However, successful pregnancy occurs in up to 35% to 80% of couples who have been evaluated for RPL, regardless of etiology and treatment (5). Couples with unexplained RPL have even higher chances of successful pregnancy.
Etiology of Recurrent Pregnancy Loss
- Chromosomal;
- Anatomic/uterine factors;
- Infectious diseases;
- Endocrine disorders;
- Immunologic disorders;
- Inherited thrombophilias;
- Environmental and other associations.
Risk factors:
- Previous pregnancy loss;
- Advancing maternal age;
- Increasing parity -- "reproductive compensation"
- Negative outcome of last pregnancy;
- Advanced gestational age at time of loss(es).
Diagnosis and Management
There is no specific number oflosses or firmly established criterion that justices evaluation for RPL or defines the scope of testing. Decisions must be individualized and consideration of the woman's age, the timing and circumstances surrounding earlier pregnancy losses, elements of the personal and family medical history, and the couple's level of anxiety must be taken into account. Clinical investigation and treatment may be considered in couples with two consecutive spontaneous losses when any of the following are present: embryonic heart activity observed in any earlier pregnancy loss; normal karyotype on products of conception from an earlier loss; age >35 years; and infertility.
Chromosomal Abnormalities
Chromosomal abnormalities occur in 64% to 88% of isolated spontaneous abortions (5)(6). Of these, 62% to 70% are autosomal trisomies, 8% to 20% are triploid or tetraploid, and 6% are related to chromosomal structural abnormalities. The prevalence of chromosomal abnormalities in RPL is at least 50% and is usually related to maternal age. In patients undergoing in-vitro fertilization (IVF), preimplantation failure is associated with chromosomal abnormalities in 67% to 85% of cases (7). Preimplantation karyotyping of embryos from recurrent abortion patients demonstrates a six-fold increased rate of monosomies. Recurrent aneuploidy (abnormal number of chromosomes) can be demonstrated in more than two thirds of subsequent pregnancies after diagnosis in a first pregnancy. The etiology of chromosomal abnormalities is not completely understood. Abnormalities that arise during the first meiotic division account for the majority of aneuploidy. The exact mechanism through which this occurs is under investigation. Genetic factors that are age-dependent, such as fragile sites on chromosomes, inversions, and translocations, account for 3.5% to 4.4% of RPL from aneuploidy. Of these, translocations are most often found in RPL. There are two major types of translocations: reciprocal, in which two segments from different chromosomes are exchanged, and Robertsonian, in which there is fusion at the centromere of two acrocentric chromosomes. Balanced translocations may result in normal carriers but can lead to unbalanced rearrangements in offspring and thus miscarriage. Pregnancy loss is more common with maternal translocations.
Frequency and Distribution of Chromosomal Abnormalities in Clinically Recognized Human Conceptions (8):
| Abnormality | Spontaneous Abortion | Stillbirth | Live Birth | Probability of Survival at Term |
|---|---|---|---|---|
| All | 50 | 5 | 0.5 | 5 |
| Trisomy 16 | 7.5 | --- | --- | 0 |
| Trisomy 13, 18, 21 |
4.5 | 2.7 | 0.14 | 15 (3, 5, 22) |
| XXX, XXY, XYY | 0.3 | 0.4 | 0.15 | 75 |
| All other trisomies | 13.8 | 0.9 | --- | 0 |
| 45XO | 8.7 | 0.1 | 0.01 | 1 |
| Triploidy 3N | 6.4 | 0.2 | --- | 0 |
| Tetraploidy 4N | 2.4 | --- | --- | 0 |
| Structural | 2.0 | 0.8 | 0.3 | 45 |
Uterine Factors
Congenital and acquired uterine anomalies account for 10% to 15% of women with RPL and are identified by sonohysterography (SHG) or hysterosalpingography (HSG) (9). Magnetic resonance imaging (MRI) may be required to accurately differentiate septate and bicornuate uteri.
Müllerian Tract Abnormalities: Congenital anomalies of the müllerian tract manifest as a spectrum of findings, from partial septa to complete duplication of the upper reproductive tract (see figure below). The septate uterus is the most common müllerian anomaly, the one most closely correlated with pregnancy loss (65% spontaneous abortion rate), and the malformation most easily and successfully corrected by hysteroscopic septoplasty. Uterine didelphys and bicornuate uterus are associated with approximately 40% spontaneous abortion rate. However, abdominal metroplasty procedures are rarely indicated for women with a uterus didelphys or a bicornuate uterus. Cervical cerclage may help to improve pregnancy outcomes in women with bicornuate uteri and in those with a unicornuate uterus or a uterus didelphys who have a history of previable delivery or exhibit progressive cervical shortening during early pregnancy.
n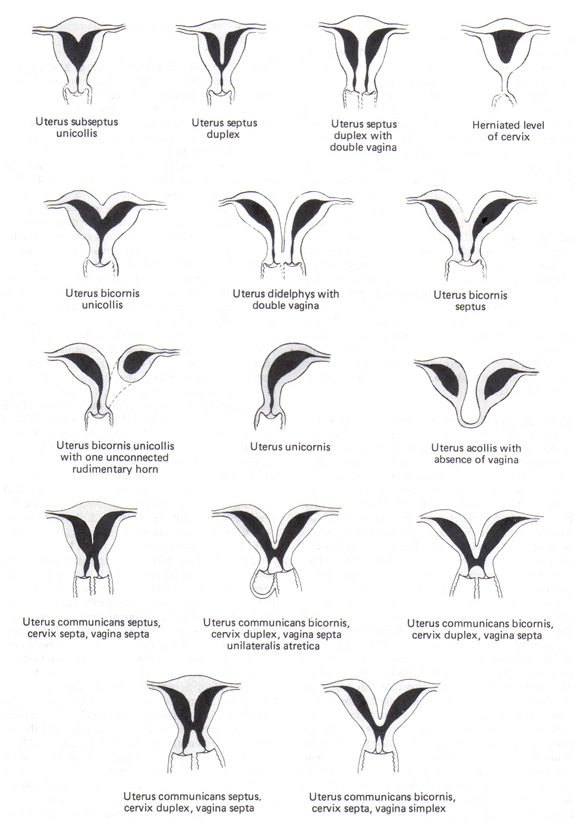
Figure 1. Congenital Uterine Anomalies
Uterine Fibroids and Other Anatomic Factors: Uterine fibroids are often identified in women with RPL, but only submucous myomas and larger intramural myomas that encroach upon or distort the uterine cavity are significant. Intrauterine adhesions are an uncommon but established cause of RPL, with improved pregnancy outcomes after hysteroscopic lysis. Lastly, although diethylstilbesterol (DES) was banned in 1971, affected women are still occasionally seen. Approximately 70% of women exposed to DES in-utero have a developmental uterine abnormality and have a two-fold increased risk of spontaneous abortion, about 24% (10). Cerclage should be considered in women with a history of DES exposure and second trimester loss or preterm delivery.
Infectious Diseases
Although infection is a frequent cause of poor pregnancy outcome latter in pregnancy, no consistent association between infections and RPL has been demonstrated. Neither Chlamydia nor Mycoplasma species, common causes of preterm birth and other complications after the first trimester, has been conclusively identified as a cause of RPL. These may, however, be associated with non-recurrent or sporadic pregnancy loss. Routine serologic testing, cervical cultures, and endometrial biopsy to detect genital infections in women with RPL are not justified. Evaluation for infection should be limited to women with cervicitis, chronic or recurrent bacterial vaginosis, or other symptoms of pelvic infection.
Endocrine Causes
Endocrine disorders are relatively uncommon cause of RPL but include: diabetes mellitus; subclinical thyroid disease; polycystic ovary syndrome (PCOS); hyperprolactinemia; and luteal phase defects. Evaluation of blood glucose and hemoglobin A1C levels is indicated for women with known or suspected diabetes mellitus, but otherwise is unwarranted. It is established that well-controlled diabetes mellitus does not increase the risk of spontaneous abortion. Thyroid disorders are easy to identify and treat and should be excluded by thyroid-stimulating hormone (TSH) measurement; even subtle abnormalities may affect pregnancy outcome although there is controversy if there is a true relationship to RPL. Insulin resistance is also associated with RPL, and PCOS has been associated with spontaneous abortion. This may be mediated via an impaired fibrinolytic response as evidenced by the fact that women with PCOS and RPL have elevated levels of plasminogen activator inhibitor-1 (PAI-1) (11). Elevated levels of PAI-1 may impede trophoblast implantation and invasion mediated by urokinase-type plasminogen activator. Other potentially embryotoxic factors found in excess in PCOS patient's follicular fluid include glucose, insulin-like growth factor-1 (IGF-1), and androgen levels. PCOS alone do not predict the risk of miscarriage. However, obesity is independently associated with pregnancy loss. Retrospective and small case-control studies have shown that therapy with metformin reduces the risk of PRL in women with PCOS from 42% to 62% to 26% to 29% (11). Larger prospective studies are necessary to confirm this association. Elevated prolactin levels have been associated with RPL, and treatment with bromocriptine has been shown to increase the likelihood of normal pregnancy.
Luteal Phase Defects
The incidence of luteal phase defect -- verified on endometrial biopsy by demonstrating a lack of progesterone effect expected in the cycle -- is the same for fertile women as for those with a history of infertility (12). Therefore, despite the reasonable theory that luteal phase defect is a potential cause of RPL, just how it is diagnosed remains uncertain. The prevalence of luteal phase defect, in which there is a relative deficiency in progesterone, is 2.5% in RPL patients (12). Although much of the available literature has been contradictory, a Cochrane review of 15 randomized controlled trials concluded that starting supplemental progestogen in the first trimester of pregnancy improves outcomes for patients with recurrent, but not isolated, pregnancy loss in the first or second trimesters (13). Given the data indicating benefit for women with second-trimester losses, by which time the placenta has taken over the role of secreting progesterone, the theory behind progesterone's effectiveness in this setting is its action as a smooth-muscle relaxant. Routes of administration and doses of progestogen varied among the reviewed studies. Some IVF studies suggest that oral progestogen is inferior to an intramuscular formulation (14). Especially with an infertility population, it is important with early supplementation not to start too early. IVF data show that an endometrium excessively accelerated may actually reduce the rate of embryo implantation. Therefore, when supplementation is provided before a positive pregnancy test result, the patient must be monitoring ovulation. For patients with RPL, certainly if it has occurred in the first trimester -- supplementation should continue at least until gestational age of 9 weeks. Implantation happens roughly a week after ovulation, but there is leeway in timing. Occasionally patients will forget to take their progestogen supplementation until later in the cycle, and no difference in pregnancy rates has been noted.
Treatment of luteal-phase defect
Progesterone vaginal suppositories: 25 mg twice a day beginning 3 days after the luteinizing hormone (LH) surge and continuing until 8 weeks' gestation;
Progesterone in oil intramuscular (IM): 12.5 mg/day IM beginning 3 days after the LH surge and continuing until 8 weeks' gestation;
Micronized progesterone: 200 mg/day by mouth beginning 3 days after the LH surge and continuing until 8 weeks' gestation;
Clomiphene citrate: 50 mg/day by mouth for 5 days beginning on days 3-5 of the menstrual cycle (no pregnancy supplementation necessary).
Immunologic Factors
Both autoimmune and alloimmune mechanisms are implicated as causes of RPL. Autoimmune disorders involve an immune response directed against a specific part of the host or self. Alloimmune disorders involve an abnormal maternal immune response to fetal or placental antigens including maternal cytotoxic antibodies, absent maternal blocking antibodies, and disturbances in natural killer cell function and distribution.
Autoimmune Diseases
Autoimmune diseases such as systemic lupus erythematosus (SLE) and the antiphospholipid syndrome (APLS) are identifiable and treatable disorders and are associated with RPL. The lupus anticoagulant and anticardiolipin antibodies, the most widely accepted antibodies of clinical use, and have been associated with a variety of medical problems, including arterial and venous thromboses, autoimmune thrombocytopenia, and fetal loss (15). In addition to fetal loss, several obstetric complications have been associated with antiphospholipid antibodies, including preeclampsia, intrauterine growth restriction, placental insufficiency, and preterm delivery.
Antiphospholipid Syndrome (APLS)
Antiphospholipid syndrome (APLS) is an autoimmune disorder defined by the presence of characteristic clinical features and specific levels of circulating antiphospholipid antibodies. Because approximately 70% of individuals with APLS are female (16), it is reasonably common among women of reproductive age. Antiphospholipid antibodies are a diverse group of antibodies with specificity for protein binding negatively charged phospholipids on cell surface. Despite the prevalence and clinical significance of APLS, there is controversy about the indications for APLS testing and the tests that should be ordered to diagnose the condition. Much of the debate results from a lack of well-designed and controlled studies on the diagnosis and management of APLS (17). Primary antiphospholipid syndrome refers to patients with APLS but no other recognized autoimmune disorders (18). However, other autoimmune conditions such as SLE often coexist with the condition. When it occurs in the setting of other autoimmune disease, it is referred to as secondary antiphospholipid syndrome (18). Definite APLS is considered to be present if at least one of the clinical criteria and one of the laboratory criteria are met (17):
Clinical Obstetric:
- Three or more consecutive spontaneous abortions before 10th week of gestation;
- One or more unexplained fetal deaths at or beyond the 10th week of gestation;
- Severe preeclampsia or placental insufficiency necessitating birth before the 34th week of gestation.
Vascular thrombosis:
- Unexplained venous thrombosis;
- Unexplained arterial thrombosis;
- Small-vessel thrombosis in any tissue or organ, without significant evidence of inflammation of the vessel wall.
Laboratory
- Anticardiolipin antibodies of IgG or IgM isotype in medium to high titers, on two or more occasions at least 6 weeks apart, measured by standardized enzyme-linked immunosorbent assay;
- Lupus anticoagulant present in plasma, on two or more occasions at least 6 weeks apart, detected according to guidelines of the International Society on Thrombosis and Hemostasis, in the following steps:
- Demonstration of a prolonged phospholipid-dependent coagulation screening test (e.g., activated partial thromboplastin time, kaolin clotting time, dilute Russell's viper venom time, dilute prothrombin time);
- Failure to correct the prolonged screening test by mixing with normal platelet-poor plasma;
- Shortening or correction of the prolonged screening test by the addition of excess phospholipids;
- Exclusion of other coagulopathies (e.g., factor VIII inhibitor, heparin) as clinically indicated.
Management of APLS during pregnancy: The goals of treatment for APLS during pregnancy are to improve maternal and fetal-neonatal outcome by reducing the risk of pregnancy loss, preeclampsia, placental insufficiency, and preterm birth and to reduce or eliminate the maternal thrombotic risks. Two recent reviews (19) have emphasized that case series and treatment trials tend to include individuals whose APLS diagnosis falls into one of two groups: those with a history of thrombotic events and those without a history. Treatment of women with APLS without a thrombotic event is controversial. A recent meta-analysis suggested that, for women with recurrent miscarriage as the clinical criteria, prophylactic heparin and low-dose aspirin may reduce pregnancy loss by 50% (20). This combined therapy appears superior to low-dose heparin alone or prednisone. For women with APLS without a history of a thrombotic event, some physicians recommend initiation of heparin before conception, although no clinical trial supports this recommendation. Most experts recommend 6-8 weeks of postpartum thromboprophylaxis in women with obstetric APLS (21). Several approaches to the peripartum management of anticoagulation therapy for these patients are available. The management at our institution is:
- Heparin (10K-20K unit subcutaneous or 0.7mg/kg/day LMWH [low molecular weight heparin] subcutaneous); and/or
- Aspirin (81 mg) + heparin (above).
Treatment may be initiated preconception to after documented viability with no detectable differences in outcome (22). Other therapies that have been suggested for treatment of pregnant women with APLS include corticosteroids and intravenous immunoglobulin (IVIG). Several case series have reported a 60-70% rate of successful pregnancies in women with APLS treated with prednisone and low-dose heparin (22). Because the efficacy of IVIG has not been proven in appropriately designed studies and the drug is extremely expensive, it is not recommended as primary therapy.
Alloimmune Disorders
The acceptance of the fetal and placental allograft by the maternal immune system results from trophoblast expression of non-immunogenic HLA-G; Fas ligand; immunosuppressive agents such as hCG, PAPP-A, and progesterone; and increased maternal cortisol levels. HLA sharing between husband and wife was theorized to result in the absence of a pregnancy-specific antibody that protected the half-foreign fetus from maternal rejection. However, all current methods for the evaluation of suspected alloimmunopathology, including HLA testing, immune cell evaluation (mixed lymphocyte culture, natural killer cell assays), and cytokine testing are investigational. Neither of the two principal immunotherapies advocated for the treatment of alloimmune disorders in women with RPL, paternal leukocyte immunization and IV immunoglobulin, have been proven to be effective (23).
Inherited Thrombophilias
Inherited thrombophilias resulting from genetic mutations in clotting factors leading to placental thrombosis have emerged as a potentially important cause of RPL (24). However, the majority of women with these mutations have completely normal reproductive outcomes. Women with more than one type of mutation or whose fetus inherits the mutation may be at a greater risk of spontaneous abortion. The indications for screening women with RPL for the growing number of recognized thrombophilias are not yet established. Currently, screening seems appropriate for women with otherwise unexplained RPL with a suspicious loss (after 10 weeks' gestation or detection of embryonic heart activity) or history of other pregnancy complications that may have resulted from thrombosis or placental insufficiency (preeclampsia, intrauterine growth restriction, placental abruption). In addition to lupus anticoagulant and anticardiolipin antibodies for diagnosis of acquired thrombophilia, screening includes tests for factor V Leiden and prothrombin gene mutation G20210A. They are the two most common inherited causes of venous thromboembolism and the thrombophilias most highly associated with adverse pregnancy outcomes (25). The prevalence of these mutations is relatively high among those of European descent (up to 15%) but very low on Asians, Africans, and Native Americans (25). Measurement of activated protein C resistance is a more global test for detection of both inherited and acquired forms of activated protein C resistance. Screening for the methylene tetrahydrofolate reductase (MTHFR) mutation, serum homocysteine and antithrombin III, protein S, and protein C deficiencies may also warrant consideration, based on past and family medical history (26).
Given the available data, there is insufficient evidence to support screening for thrombophilia as part of the initial evaluation of RPL. In addition, it is unknown whether treatment will improve pregnancy outcome in these women. Most early pregnancy losses are due to aneuploidy and ideally all women with RPL (and fetal death) should have complete workups, including karyotype of losses to exclude other causes before attributing losses to thrombophilias. Thrombophilias are convincingly associated with an increased relative risk for fetal death, but not with recurrent first-trimester loss. Similarly, treatment may help those with thrombophilia and fetal death but treatment has not been tested in women with recurrent first-trimester loss and thrombophilia. Preliminary data suggest that combined treatment with aspirin and heparin may improve pregnancy outcomes in women with RPL and thrombophilia but prospective, controlled trials are required.
Environmental and Other Associations
Smoking increases the risk of spontaneous abortion and should be discouraged. Alcohol consumption exceeding two drinks per day and high caffeine consumption (more than 300 mg/day) may increase risk for pregnancy loss and best avoided. Environmental toxins such as heavy metals (mercury, lead), organic solvents (e.g. perchlorethlene or dry cleaning solvent), ionizing radiation, and anesthetic gases have been implicated as causative agents for spontaneous abortion (27). Exposure to video terminals, electric blankets, or heated waterbeds is not associated with an increased risk of RPL. Exercise programs do not increase risk, and bed rest will not decrease the risk of RPL.
| Recurrent Pregnancy Loss Checklist | ||
|---|---|---|
| Laboratory Tests | Date | Result |
| SHG or HSG | ||
| Karyotype ♀ ♂ |
||
| Fasting insulin/glucose; HbA1C | ||
| MTHFR (C677T or A1298C) | ||
| Homocysteine | ||
| PG20210A | ||
| Factor V Leiden | ||
| Protein C | ||
| Protein S | ||
| Antithrombin III | ||
| Lupus anticoagulant | ||
| Anticardiolipin antibodies IgG IgM |
||
| β2- glycoprotein-I antibodies IgG IgM |
||
| TSH | ||
| Thyroid peroxidase antibody | ||
| Prolactin | ||
| Tissue transglutaminase IgA/total IgA | ||
Abbreviations: SHG -- sonohysterography; HSG -- hysterosalpingography; MTHFR -- methylene tetrahydrofolate reductase; TSH -- thyroid stimulating hormone;
Follow-Up
After complete evaluation and successful treatment, couples who have experienced spontaneous abortion are often afraid to become pregnant again. These patients need intensive physician support during their first trimester. It is useful to inform couples that the risk of spontaneous abortion decreases as the duration of pregnancy increases: observation of a gestational sac (12%), yolk sac (8%), embryonic crown-rump length increases (>5mm, 7%; 6-10 mm, 3%; >10 mm, <1%). The observation of embryonic cardiac activity by 6 weeks' gestation reduces the risk of spontaneous abortion to 3% - 5%. However, the incidence of subsequent spontaneous abortion is higher when there are other abnormal sonographic findings: slow or late appearing heart activity, size and/or date discrepancies, or subchorionic hematoma. Finally, the prognostic value of embryonic heart activity decreases with increasing maternal age from spontaneous abortion rate of less than 5% in women under age 35 to 29% in women aged 40 and over.
Summary
Spontaneous abortion is the most common complication of pregnancy. 40% to 50% of conceptuses are lost before expected menses, and 30% to 35% are lost at or following missed menses. Traditionally recurrent pregnancy loss (RPL) refers to the loss of three or more consecutive pregnancies; however, many clinicians will begin the evaluation of RPL after two losses, because the risk of a third loss after two miscarriages is approximately 30%, whereas the risk after three losses is about 33%. This approach may be especially useful in older women. The risk of miscarriage is lower in women who carry at least one pregnancy to term, whereas a history of miscarriage increases the chance of miscarriage in subsequent pregnancies. Maternal age is also a strong predictor of recurrent loss. The major causes of RPL are chromosomal, anatomic/uterine, endocrine, immunologic, and thrombophilic. The evaluation of couple with RPL should include the following: parental karyotypes; uterine evaluation with sonohysterography (SHG), HSG, and/or hysteroscopy; evaluation of APLS: lupus anticoagulant, anticardiolipin antibodies, antibodies to β2-glycoprotein-I; a workup for inherited thrombophilias if the losses have occurred after 10 weeks; and serum prolactin level. The workup of RPL is negative in more than 50% of patients. However, successful pregnancy occurs in up to 35% to 80% of couples who have been evaluated for RPL, regardless of the etiology and treatment. It is important to remember that couples who are being evaluated for RPL have high levels of depression and stress. Some studies have indicated that psychological support may decrease the rates of unexplained miscarriage. Finally, patients should be reassured that even without treatment, successful pregnancy occurs in the majority of cases.
References
- Jaslow CR, Carney JL, Kutteh WH. Diagnostic factors identified in 1020 women with two versus three or more recurrent pregnancy losses. Fertil Steril 2010;93(4):1234-1243
- Speroff L, Glass R, Kase N. Clinical Gynecologic Endocrinology and Infertility. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 1999
- ACOG Practice Bulletin. Management of recurrent pregnancy loss. Number 24, February 2001. (Replaces Technical Bulletin Number 212, September 1995). American College of Obstetricians and Gynecologists. Int J Gynaecol Obstet 2002;78:179-185
- Cramer DW, Wise LA. The epidemiology of recurrent pregnancy loss. Semin Reprod Med 2000;18:331-339
- Sfakianaki AK, Lockwood CJ. In High Risk Obstetrics -- the requisites in Obstetrics and Gynecology; Recurrent pregnancy loss. 2008; p.147. Publisher: Mosby, Elsevier
- Dhont M. Recurrent miscarriage. Curr Women's Health Rep 2003;3:361-366
- Baart EB, Martini C, van den Berg I, et al. Preimplantation genetic screening reveals a high incidence of aneuploidy and mosaicism in embryos from young women undergoing IVF. Hum Reprod 2006;21:223-232
- Kodaman PH. Recurrent pregnancy loss. Yale CME Frontiers of reproduction at Yale 2011: fertility and women's health. April, 29, 2011
- Propst Am, Hill JA. Anatomic factors associated with recurrent pregnancy loss. Semin Reprod Med 2000;18:341-350
- Saravelos SH, Cocksedge K, Li TC. The pattern of pregnancy loss in women with congenital uterine anomalies and recurrent miscarriage. Reprod Biomed 2010;20(3):416-422
- Lee RM, Silver RM. Recurrent pregnancy loss: summary and clinical recommendations. Semin Reprod Med 2000;18:433-440
- Coutifaris C, Myers ER, Guzick DS, et al. Histological dating of timed endometrial biopsy tissue is not related to fertility status. Fertil Steril 2004;82(5):1265-1272
- Haas DM, Ramsey PS. Progestrogen for preventing miscarriage. Cochrane Database Syst Rev. 2008;16(2): CD003511
- Penzias AS. Luteal phase support. Fertil Steril 2002;77(2):318-323
- Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med 2002;346:752-763. (Level III)
- Lockshin MD. Antiphospholipid antibody. Babies, blood clots, biology. JAMA1997;277:1549-1551. (Level III)
- ACOG Practice Bulletin. Antiphospholipid syndrome. Number 68; November 2005
- Levine JS, Branch DW, Rauch J. The antiphospholipid syndrome. N Engl J Med 2002;346:752-763. (Level III)
- Derksen RH, Khamshta MA, Branch DW. Management of the obstetric antiphospholipid syndrome. Arthritis Rheum 2004;50:1028-1039. (Level III)
- Ziakas PD. Pavlou M, Voulgarelis M. Heparin treatment in antiphospholipid syndrome with recurrent pregnancy loss. Obstet Gynecol 2010115:1256-1262
- Empson M, Lassere M, Craig JC, Scott JR. Recurrent pregnancy loss with antiphospholipid antibody: a systematic review of therapeutic trials. Obstet Gynecol 2002;99:135-144. (Level III)
- Bates SM, Greer IA, Hirsh J, et al. Use of antithrombotic agents during pregnancy: the Seventh ACCP Conference in Antithrombotic and Thrombolytic Therapy. Chest 2004;126(suppl 3):627S-644S. (Level III)
- Porter TF, Scott JR. Alloimmune causes of recurrent pregnancy loss. Semin Reprod Med 2000;18:341-350
- Rey E, Kahn SR, David M, et al. Thrombophilic disorders and fetal loss: a meta-analysis. Lancet 2003;361:901-908. (Meta-Analysis)
- Lissalde-Lavigne G, Fabbro-Peray P, Cochery-Nouvellon E, et al. Factor V Leiden and prothrombin G20210A polymorphisms as risk factors for miscarriage during a first intended pregnancy: the matched case control 'NOHA first' study. J Thromb Haemost 2005;3:2178-2184. (Level II-2)
- ACOG Practice Bulletin. Inherited thrombophilias in pregnancy. Number 111, April 2010
- Rai R, Regan L. Recurrent miscarriage. Lancet 2006;368:601-605
Published: 19 May 2014
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


