HELLP Syndrome - Diagnosis and Management
Dr. Baha M. Sibai
Professor and Chairman
Department of Obstetrics and Gynecology
University of Cincinnati, Ohio (USA)
Intravascular hemolysis, elevated liver function tests and low platelets counts (thrombocytopenia) also known as HELLP syndrome has been recognized as a complication of severe preeclampsia and eclampsia for many decades. The purpose of this document is to describe the pathogenesis, diagnosis and management of this syndrome. The presence of this syndrome is associated with increased risk of adverse outcome for both mother and fetus. This review will explain the controversies surrounding the diagnosis and management of this syndrome. Recommendations for the counseling of these women are also provided based on the results of recent studies.
Diagnostic Criteria:
The diagnostic criteria used for HELLP syndrome are variable and inconsistent. The term HELLP syndrome (H = hemolysis; EL = elevated liver enzymes; and LP = low platelets) is the collection of laboratory abnormalities described in patients with severe preeclampsia and eclampsia. The classic findings of microangiopathic hemolysis include abnormal peripheral smear (schistocytes, burr cells, echinocytes), elevated serum bilirubin (indirect form), low serum haptoglobin levels, elevated lactate dehydrogenase (LDH) levels (threshold of 180 - 600 U/L), and significant drop in hemoglobin levels. Low platelet count is the third abnormality required to establish the diagnosis of HELLP syndrome. The reported cut-off values range from 75,000/mm3 to 279,000/mm3. Many authors have used total LDH (usually more than 600 U/L) as a diagnostic criterion for hemolysis. There are 5 different isoforms of LDH, and only 2 of them, LDH1 and LDH2, are released from ruptured red blood cells. In the majority of women with severe preeclampsia - eclampsia, the elevation in total LDH is probably caused mostly by liver ischemia. Therefore, many authors advocate that elevated bilirubin values (indirect form), abnormal peripheral smear, or a low serum haptoglobin level should be part of the diagnostic criteria for hemolysis.
It is important to recognize that both maternal and perinatal outcomes are expected to be substantially worse in those patients whom HELLP syndrome develops in the second trimester and who require emergency cesarean delivery because of non-reassuring fetal testing than in women in whom severe preeclampsia develops at term and have spontaneous vaginal delivery and subsequently had the diagnosis of HELLP syndrome made because of frequent evaluation of liver enzymes and platelet count during labor and in immediate postpartum period.
Clinical Findings:
One of the major problems with early detection of HELLP syndrome lies in its clinical presentation, because patients may present with non-specific symptoms or subtle signs of preeclampsia. Patients with this syndrome may present with various signs and symptoms, none of which are diagnostic of preeclampsia and all of which may be found in patients with severe preeclampsia-eclampsia without HELLP syndrome. Patients frequently presents with right upper quadrant or epigastric pain, nausea, or vomiting ranging in frequency from 30% to 90%. Most patients will give a history of malaise for the past few days before presentation, and some will have non-specific viral-syndrome-like symptoms. Headaches are reported by 33-61% of the patients whereas visual changes are reported in approximately 17%. A subset of patients with HELLP syndrome may present with symptoms related to thrombocytopenia such as bleeding from mucosal surfaces, hematuria, petechial hemorrhages or ecchymosis.
Although the majority of patients will have hypertension (82-88%), it may be only mild in 15-50% of the cases, and absent in 12-18%. The majority of the patients (86-100%) will have proteinuria, however it is reported to be absent in 13% of cases in some large series.
Differential Diagnosis:
The presenting symptoms, clinical findings, and many of the laboratory findings in women with HELLP syndrome overlap with a number of medical syndromes, surgical conditions, and obstetric complications. Because some patients with HELLP syndrome may present with gastrointestinal, respiratory, or hemorrhagic symptoms in association with elevated liver enzymes or low platelets in the absence of hypertension or proteinuria, many cases of HELLP syndrome will initially be misdiagnosed as upper respiratory infection, hepatitis, cholecystitis, pancreatitis, acute fatty liver of pregnancy, or immune thrombocytopenic purpura. In addition, in some patients preeclampsia may be super-imposed on one of these disorders, further contributing to the diagnostic difficulty. Therefore, it is very important to make an effort to attempt to identify an accurate diagnosis given that management strategies may differ among these conditions. The clinical and laboratory findings that mimic the HELLP syndrome are: acute fatty liver of pregnancy (AFLP), thrombotic thrombocytopenic purpura (TTP), hemolytic uremic syndrome (HUS), immune thrombocytopenic purpura (ITP), systemic lupus erythematosus (SLE), antiphospholipid syndrome (APS), cholecystitis, fulminating viral hepatitis, acute pancreatitis, disseminated herpes simplex, hemorrhagic or septic shock.
Maternal and Perinatal Outcome:
The presence of HELLP syndrome is associated with an increased risk of maternal death (1%) and increased rates of maternal morbidities such as pulmonary edema (8%), acute renal failure (3%), disseminated Intravascular coagulopathy (DIC, 15%), abruptio placentae (9%), liver hemorrhage or failure (1%), adult respiratory distress syndrome (ARDS), sepsis, and stroke (<1%). Pregnancies complicated by HELLP syndrome are also associated with increased rates of wound hematomas and the need for transfusion of blood and blood products. The development of HELLP syndrome in the postpartum period also increases the risk of renal failure and pulmonary edema. The reported perinatal death rate in recent series ranged from 7.4% to 20.4%. The high perinatal death rate is mainly experienced at very early gestational age (less than 28 weeks), in association with severe fetal growth restriction or abruptio placentae. It is important to emphasize that neonatal morbidities in these pregnancies are dependent on gestational age at time of delivery and they are similar to those in preeclamptic pregnancies without HELLP syndrome. The rate of pre-term delivery is approximately 70%, with 15% occurring before 28 weeks of gestation. As a result, these infants have a high rate of acute neonatal complications such as respiratory distress syndrome, broncho-pulmonary dysplasia, intracerebral hemorrhage, and necrotizing enterocolitis.
Management of HELLP Syndrome:
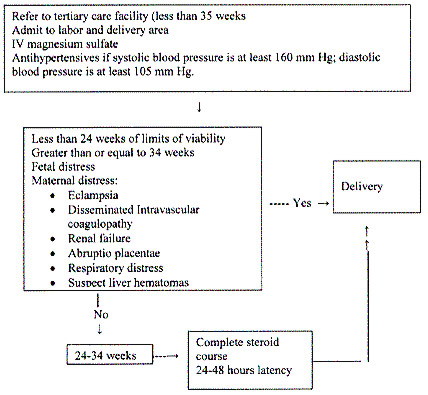
The clinical course of women with true HELLP syndrome is usually characterized by progressive and sometimes sudden deterioration in the maternal condition. There is a consensus of opinion that prompt delivery is indicated if the syndrome develops beyond 34 weeks of gestation, or earlier if there is multi-organ dysfunction, DIC, liver infarction or hemorrhage, renal failure, suspected abruptio placentae, or non-reassuring fetal status.
There is considerable disagreement about the treatment of women with HELLP syndrome at or before 34 weeks of gestation when the maternal condition is stable except for mild-to-moderate abnormalities in blood test results and a reassuring fetal condition. In such patients our recommendation is the administration of corticosteroids to accelerate fetal lung maturity followed by delivery after 24 hours. Some of the measures used in this group is one or more of the following: bed rest, antihypertensive agents, chronic parenteral magnesium sulfate, antithrombotic agents (low-dose aspirin, dipyridamole), plasma volume expanders (crystalloids, albumin, fresh frozen plasma), and steroids (prednisone, dexamethasone, or betamethasone).
The expectant management in very select group of patients with HELLP syndrome before 34 weeks of gestation is possible. Despite pregnancy prolongation in some of these cases, the overall perinatal outcome was not improved compared with cases at similar gestational age who were delivered within 48 hours after the diagnosis of HELLP syndrome. Therefore, such management remains experimental in absence of randomized trials.
Corticosteroids Use To Improve Pregnancy Outcome:
It is well established that antenatal glucocorticoid therapy reduces neonatal complications and neonatal mortality in women with severe preeclampsia at or before 34 weeks of gestation. The recommended regimens of corticosteroids for the enhancement of fetal maturity are betamethasone (12 mg intramuscularly every 24 hours, 2 doses) or dexamethasone (6 mg intramuscularly every 12 hours, 4 doses). These regimens have been identified as the most appropriate for this purpose because they readily cross the placenta and have minimal mineralocorticoid activity. However, it is unclear whether the same or different regimens are safe and effective in women with HELLP syndrome. Corticosteroids have been suggested as safe and effective drugs for improving maternal and neonatal outcome in women with HELLP or partial HELLP syndrome. The National Institutes of Health Consensus Development Panel recommends standard-dose corticosteroids use to improve perinatal outcome when used in women with HELLP syndrome before 34 weeks of gestation. In addition, in some of these women, there is transient improvement in maternal platelet counts, which makes them eligible to receive epidural anesthesia.
There is also some evidence suggesting improved laboratory values with the use of higher doses of dexamethasone in women with postpartum HELLP syndrome. The doses of dexamethasone considered "high dose" in these studies was 10 mg of dexamethasone administered intravenously every 6 to 12 hours for 2 doses followed by 5-6 mg intravenous doses given 6 to 12 hours later for 2 additional doses. Routine use of the high-dose dexamethasone to improve maternal outcome in women with HELLP syndrome beyond 34 weeks of gestation or in the postpartum period remains experimental.
Sibai, Diagnosis and Management of HELLP syndrome. Obstet Gynecol 2004.
Intrapartum Management:
The presence of HELLP syndrome is not an indication for immediate cesarean delivery. Such an approach might prove detrimental for both mother and fetus. The decision to perform cesarean delivery should be based on fetal gestational age, fetal conditions, presence of labor, and cervical Bishop Score. Our suggestion is to recommend elective cesarean delivery for all women with HELLP syndrome before 30 weeks of gestation who are not in labor and whose Bishop score is below 5. We also recommend elective cesarean delivery to those with HELLP syndrome plus fetal growth restriction and/or oligohydramnios if the gestational age is below 32 weeks in the presence of an unfavorable cervical Bishop score.
Patient having labor or rupture of membranes are allowed to deliver vaginally in the absence of obstetric complications. When labor is indicated, it is initiated with either oxytocin infusions or prostaglandins in all patients with a gestational age at or above 30 weeks, irrespective of the extent of cervical dilation or effacement. A similar approach is used for those at or before 30 weeks of gestation if the cervical Bishop score is at least 5.
Maternal pain relief during labor and delivery can be provided by intermittent use of small doses of systemic opioids. Local infiltration anesthesia can be used for all vaginal deliveries in case of episiotomy or laceration repair. The use of pudendal block is contraindicated in these patients because of the risk of bleeding and hematoma formation into this area. Epidural anesthesia is also contraindicated, particularly if the platelet count is less than 75,000/mm3. Therefore, general anesthesia is the method of choice for cesarean delivery in such patients. The impact of glucocorticoid administration on the rate of epidural anesthesia use has been studied by some researchers with partial HELLP syndrome who had a platelet count below 90,000/mm3 before steroid administration. It has been seen that administration of corticosteroids in these patients increased the rate of epidural anesthesia use, particularly in those who achieved a latency period of 24 hours before delivery. Platelet transfusions are indicated either before or after delivery in all patients with HELLP syndrome in the presence of significant bleeding (ecchymosis, bleeding from gums, oozing from puncture sites, wound, intraperitoneal etc.), and in all those with a platelet count of less than 20,000/mm3. Repeated platelet transfusions are not necessary because of the short-life of the transfused platelets in such patients. Correction of thrombocytopenia is also important before any surgery. Our policy is to administer 6 U of platelets in all patients with a platelet count less than 40,000/mm3 before intubation if cesarean delivery is needed. Generalized oozing from the surgical site can occur during surgery or in the immediate postpartum period because of the continued drop in platelet count in some of these patients. The risk of hematoma formation at these sites is approximately 20%. Therefore, our policy is to use a subfascial drain and to keep the skin incision open for at least 48 hours in all patients requiring cesarean delivery.
Postpartum Management:
After delivery, patients with HELLP syndrome should receive close monitoring of vital signs, fluid intake and output, laboratory values, and pulse oximetry for at least 48 hours. Our policy is to continue intravenous magnesium sulfate prophylaxis for 48 hours, and to use antihypertensive drugs if the systolic blood pressure is at least 155 mm Hg or if the diastolic pressure is at least 105 mm Hg. In general, the majority of patients will show evidence of resolution of the disease process within 48 hours after delivery. However, some patients, especially those with abruptio placentae plus DIC, those with severe thrombocytopenia (platelet count less than 20,000/mm3), and those with severe ascites or significant renal dysfunction may show delayed resolution or even deterioration in their clinical conditions. Such patients are at risk of the development of pulmonary edema from transfusion of blood and blood products, fluid mobilization, and compromised renal function. These patients are also at risk of acute tubular necrosis and need for dialysis, and may require intensive monitoring for several days.
The clinical and laboratory findings of HELLP syndrome may develop for the first time in the postpartum period. In these patients, the time of onset of the manifestations ranges from a few hours to 7 days, with majority developing within 48 hours postpartum. Hence, all postpartum women and healthcare providers should be educated and be aware of the signs and symptoms of HELLP syndrome. The treatment of patients should be similar to that in the antepartum period, including the use of magnesium sulfate.
Subsequent Pregnancy and Patient Counseling:
Pregnancies complicated by HELLP syndrome may be associated with life-threatening complications for both the mother and her infant. These women have increased risk of preeclampsia in subsequent pregnancies. In general, the rate of preeclampsia in subsequent pregnancies is approximately 20% with significantly higher rates if the onset of HELLP syndrome was in the second trimester. The rate of recurrent HELLP syndrome ranges from 2% to 19%. Our policy is to give these women a recurrence risk of less than 5%. Therefore, they require close monitoring in subsequent pregnancies. The development of HELLP syndrome with or without renal failure does not affect long-term renal function.
Summary:
Despite the voluminous literature on this subject, there is an urgent need for a uniform definition and diagnostic criteria for HELLP syndrome. Pregnancies complicated by this syndrome require a well-formulated management plan. The presence of HELLP syndrome is associated with life-threatening maternal and fetal complications. The potential benefits of expectant management in those remote from term and the use of corticosteroids to improve maternal outcome remain experimental.
Acknowledgement: We at Women's Health & Education Center, are grateful to Dr. Baha M. Sibai for sharing his priceless work and research in high-risk pregnancies with us. We are honored to have him as a friend and a teacher with us. His pioneer work in this area has helped millions of women worldwide. We are looking forward to work with him for many more years to come.
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


