Clinical Management of Endometriosis
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC).
Endometriosis is a chronic and recurrent reproductive disorder with variable clinical presentations. Management varies depending on the patients age, symptoms, extent of the disease, reproductive goals, treatment risks, side effects, and cost considerations. It is relatively common chronic gynecologic disorder that usually presents with chronic pelvic pain or infertility. The presence of viable, estrogen-sensitive, endometrial-like glands and stroma associated with an inflammatory response outside the uterus is globally referred to as endometriosis. Although nine decades have passed since it was first described by Sampson, endometriosis is still a puzzling disorder because of its diverse clinical presentations and controversial etiologic origins. Once endometriosis is diagnosed in a reproductive-aged woman, it should be considered a chronic disease with multiple recurrent episodes of pain, infertility, and longer symptom-free intervals. The estimated annual cost for treatment of endometriosis is more than $ 20 billion. Three subtypes of endometriosis are differentiated by gross and microscopic inspection consisting of endometriomas (ovarian cysts), superficial endometriotic implants (focus of disease primarily on the peritoneum), and deeply infiltrating endometriosis (rectovaginal nodules). Each form may have its own etiology or share origins with the other forms of this common chronic gynecologic malady.
The purpose of this document is to review the agents used in the medical management of endometriosis and discuss the use of assisted reproduction technique (ART) for patients with endometriosis who desire pregnancy. The etiology, the relationship between the extent of disease and the degree of symptoms, the effect on fertility, and the most appropriate treatment of endometriosis remain incomplete. This review also presents the evidence, including risks and benefits, for the effectiveness of medical and surgical therapy for adult women who are symptomatic with pelvic pain or infertility or both. The latest approaches using the variety of available medical and surgical treatments are discussed as they specifically relate to the adolescent population. Endometriosis is a relatively common chronic gynecologic disorder that usually presents with chronic pelvic pain or infertility. The societal effect of this disorder is enormous both in monetary costs and in quality of life.
Incidence:
This enigmatic disease affects 6-10% of reproductive aged women and has been found in women between the ages of 12 and 80 years (1). The average age of diagnosis is approximately 28 years (2). The prevalence rate of 38% (range, 20-50%) is found in infertile women and in 71-87% of women with chronic pelvic pain (3). Contrary to much speculation, there are no data to support the view that the incidence of endometriosis is increasing, although improved recognition of endometriotic lesions may have led to an increase in the rate of detection. There also appears to be no particular racial predisposition to endometriosis. A familial association of endometriosis has been suggested, and patients with an affected first-degree relative have nearly a 7 to10-fold increased risk of developing endometriosis (4). The proposed inheritance is characteristic of a polygenic-multifactorial mechanism. A number of genetic polymorphisms have been identified. In the United States, the estimated costs of diagnosing endometriosis and treating associated pain and infertility totaled $ 22 billion in 2002 (7). Given the known recurrence rate for endometriosis over time, without medical therapy, more women will likely have their endometriosis track toward disease progression rather than resolution. In addition, pain sensation does not necessarily correlate with identification of lesions. The mechanisms responsible for ongoing symptom expression likely are complex and multifactorial.
Pathophysiology:
Most experts would concur that the disease is multifactorial in etiology. Longstanding postulates include retrograde menstruation with refluxed menstruum implanting on pelvic structures, coelomic pluripotential mesothelial cells lining the peritoneum undergoing metaplasia into endometrial tissue, and implantation of cells through hematogenous or lymphatic embolization. However, a conundrum persists: why does the disease develop in some women and in others it does not? A greater understanding of the molecular mechanisms associated with endometriosis has enhanced the traditional theories. Recent advances regarding etiology of endometriosis must be applied judiciously in clinical practice to achieve optimal treatment outcomes. Retrograde transplanted endometrial tissue and cells attach to peritoneal surfaces, establish a blood supply, and invade nearby structures (5). They are infiltrated by sensory, sympathetic, and parasympathetic nerves and elicit an inflammatory response. Endometriotic implants secrete estradiol (E2) as well as prostaglandin E2 (PGE2), agents that attract macrophages (monocyte chemotactic protein 1[MCP-1]), neurotrophic peptides (nerve growth factor [NGF]), enzymes for tissue remodeling (matrix metalloproteinases [MMPs]) and tissue inhibitors of MMPs (TIMPs), and proangiogenic substances such as vascular endothelial growth factor (VEGF) and interleukin-8 (6). Lesions secrete haptogobin, which decreases macrophage adhesion and phagocytic function. Lesions and activated macrophages, which are abundant in the peritoneal fluid in women with endometriosis, also secrete proinflammatory cytokines (interleukin-1β, interleukin-8, interleukin-6, and tumor necrosis factor-α [TNF-α]). Local (and systemic) estradiol can stimulate lesion production of PGE2, which can activate pain fibers, enhance neuronal invasion of lesions by stimulating production of NGF and other neurotrophins, and promote sprouting of nociceptors that contribute persistent inflammatory pain and inhibit neuronal apoptosis. Endometrial bleeding factor (EBAF) is not rightly expressed and may contribute to uterine bleeding. Infertility results from the toxic effects of the inflammatory process on gametes and embryos, compromised fimbrial function, and ectopic endometrium that is resistant to the action of progesterone, and thus the endometrium is inhospitable to an implanting embryo (7). HoxA10 and HoxA11 genes and αVβ3 integrin are not up-regulated by progesterone, and thus the endometrium is inhospitable to an implanting embryo. Endocrine-disrupting chemicals can contribute to progesterone resistance and perhaps immune dysfunction (7). ERFFI1 (ErbB receptor feedback inhibitor 1) is constitutively expressed and there is excess mitogenic signaling.
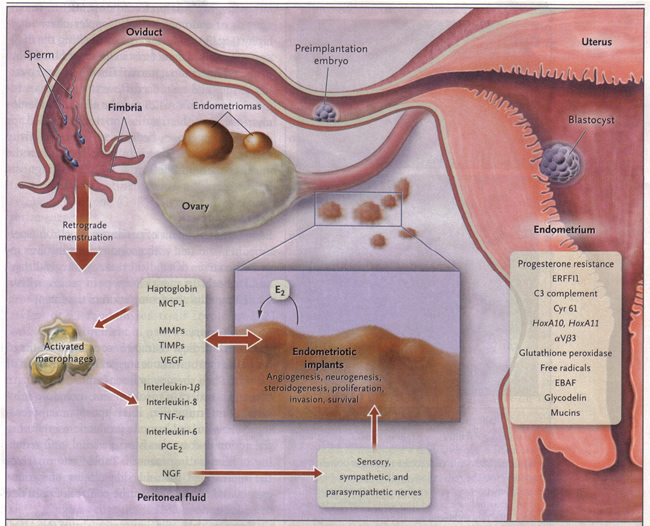
Figure 1. Pathophysiology of Pain and Infertility Associated with Endometriosis
Mechanism of Pain: Many studies have noted the presence of increased proinflammatory cytokines and growth factors in endometriosis that are closely related to pain sensation (i.e. nerve growth factor, prostaglandin, estradiol) (8). This may explain why minimal endometriosis may still cause significant pain. Current evidence indicates that endometriosis is a hyperalgesic state that results from augmented pain processing that is dictated by the way the spinal cord and the brain process pain from the lesions and other sensory information. Peripheral nerve fibers supplying endometriotic lesions could sensitize spinal segment neurons and eventually lead to a central nervous system sensitization, resulting in an exaggerated central nervous system response or phantom-like endometriotic pain, or both, despite ablation of lesions (9). This multidimensional symptomatology of endometriosis warrants multidisciplinary management strategies.
Mechanism of Subfertility: Advanced cases of endometriosis often entail severe adhesive disease that could pose an obvious impairment to fertility by reducing tubo-ovarian motility, which ultimately impedes pick-up function. Whether milder forms of the disease even cause subferility is not certain. Infertile women with minimal to mild endometriosis had lower serum antimullerian hormone levels on day 3 than infertile patients with tubal obstruction (10). In minimal or mild endometriosis, the biologic mechanism causing infertility remains elusive. Once again, an enhanced immune response may be the culprit by impairing fertility in early-stage disease (11). A hostile peritoneal environment may lead to impaired sperm function through increased sperm DNA damage, and a compromised oocytes cytoskeleton. Another theory places the onus on an endometrial defect based on reports of decreased expression for several biomarkers of implantation. Unfortunately, endometriosis is associated with an increased risk for preterm birth, antepartum complications and preeclampsia.
Diagnosis and Classification
The clinical manifestations of endometriosis are variable and unpredictable in both presentation and course. Dysmenorrhea, chronic pelvic pain, dyspareunia, uterosacral ligament nodularity, and an adnexal mass (either symptomatic or asymptomatic) are among the well-recognized manifestations. A significant number of women with endometriosis remain asymptomatic. The definitive diagnosis of endometriosis only can be made by histology of lesions removed at surgery. Neither serum markers nor imaging studies have been able to supplant diagnostic laparoscopy for the diagnosis of endometriosis. The histologic appearance consists of endometrial glands and stroma with varying amounts of inflammation and fibrosis (12).
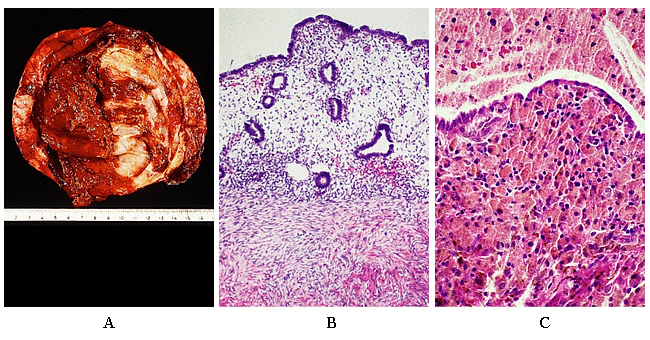
Figure 2. A: Inner surface of cyst in a case of ovarian endometriosis. The color is typically brown. B: Ovarian Endometriosis. In this area endometrial tissue faithfully reproduces the appearance of normal endometrium, in terms of both glands and stroma. C: Common appearance resulting from repeated hemorrhage and accumulation of hemosiderin-laden macrophages.
American Society for Reproductive Medicine; Revised Classification of Endometriosis
Numerous classification schemes have been proposed to describe endometriosis by anatomic and severity of disease. The American Society for Reproductive Medicine (ASRM) classification, which is the most commonly used system, was revised for the third time in 1996 but still has limitations (13). The system is not a good predictor of pregnancy after treatment despite adjustments to the point scores and cut-points for disease stage. The ASRM system does not correlate well with the symptoms of pain and dyspareunia or infertility. The value of the ASRM revised system is in uniform recording of operative findings and perhaps for comparing the results of various therapies.
| Peritoneum | Endometriosis | <1 cm | 1-3 cm | >3 cm |
|---|---|---|---|---|
| Superficial | 1 | 2 | 4 | |
| Deep | 2 | 4 | 6 | |
| Ovary | R-Superficial | 1 | 2 | 4 |
| Deep | 4 | 16 | 20 | |
| L-Superficial | 1 | 2 | 4 | |
| Posterior cul-de-sac obliteration |
Partial / Complete | |||
| 4 | 40 | |||
| Ovary | Adhesions | <1/3 Enclosure |
1/3-2/3 Enclosure |
>2/3 Enclosure |
| R-Filmy | 1 | 2 | 4 | |
| Dense | 4 | 8 | 16 | |
| L-Filmy | 1 | 2 | 4 | |
| Dense | 4 | 8 | 16 | |
| Tube | R-Filmy | 1 | 2 | 4 |
| Dense | 4* | 8* | 16 | |
| L-Filmy | 1 | 2 | 4 | |
| Dense | 4* | 8* | 16 |
*if the fimbriated end of the fallopian tube is completely enclosed, change the point assignment to 16. Denote appearance of superficial implant types as red [(R), red, red-pink, flame-like, vesicular blobs, clear vesicles], white [(W), opacifications, peritoneal defects, yellow-brown], or black [(B), black, hemosiderin deposits, blue]. Denote percent of total described as R_%, W_%, and B_%. Total should equal 100%.
- Stage I (minimal) 1-5;
- Stage II (mild) 6-15;
- Stage III (moderate) 16-40;
- Stage IV (Severe) >40
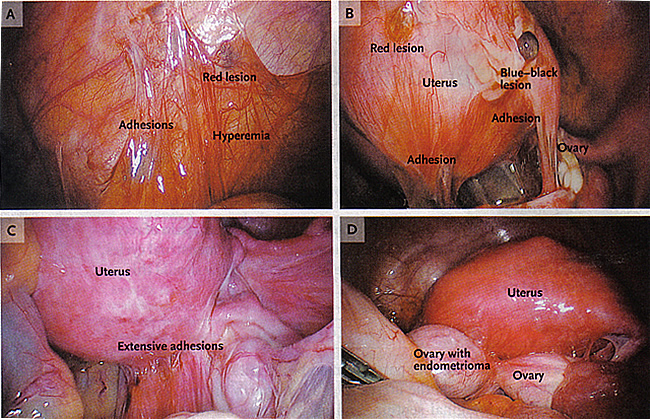
Figure 3. A: Shows an endometriotic implant (red lesion), adhesions, and hyperemia in the peritoneum. B: Peritoneal implants, including red and blue-black lesions and adhesions. C: Extensive adhesions distorting the normal pelvic anatomy. D: An endometrioma adherent to the posterior uterus and distending the ovarian capsule.
Management Goals of Endometriosis
Endometriosis has several unique biological properties. It behaves as a chronic and recurrent disease because of microscopic implants that continue to be active after surgical treatment. Many patients have quiescent disease with rare episodes of pain, while others have frequent, recurrent pain. These differing symptom patterns are often not correlated with lesion size or extent of disease. Current approaches for managing endometriosis are symptom oriented, aimed mainly at treating chronic pelvic pain and infertility. Management protocols may also be targeted at slowing disease progression or preventing recurrence. Medical, surgical, or a combined approach can be chosen. In addition, assisted reproductive techniques (ART) are frequently used to treat endometriosis-associated infertility. Treatment strategies for women with endometriosis vary according to the patients age, treatment goals, parity, extent of the disease, and menopausal status. For young patients with mild disease not desiring children, it is advisable to use medical suppression to control symptoms and minimize surgical interventions. For women with severe disease who desire fertility, surgery followed by ART is a reasonable option. Lastly, the management of surgically menopausal patients with symptomatic endometriosis requires a different approach, in which off label use of new medical therapies, such as aromatase inhibitors, may be considered.
Medical Management of Endometriosis-Associated Pain
Chronic pelvic pain accounts for 10% of outpatient gynecologic visits (14). A complete medical, surgical, social, and family history should be obtained from patients who present with this symptom, and they should undergo a physical examination that includes a pelvic examination. Focal pain and tenderness on pelvic examination is associated with pelvic disease in 97% of patients and with endometriosis in 66% of patients (14). Pelvic pain due to endometriosis is usually chronic (lasting >6 months) and is associated with dysmenorrhea (in 50% to 90% of cases), dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous, and it can be dull, throbbing, or sharp, and exacerbated by physical activity. Bladder- and bowel-associated symptoms (nausea, distention, and early satiety) are typically cyclic. Pain often worsens over time and may change in character; infrequently, women report burning or hypersensitivity, symptoms that are suggestive of a neuropathic component. Symptoms overlap with those of several other gynecologic conditions (e.g. pelvic inflammatory disease, pelvic adhesions, ovarian cysts or masses, leiomyomata, and adenomyosis) and non-gynecologic conditions and factors (e.g. irritable bowel syndrome, inflammatory bowel disease, interstitial cystitis, myofascial pain, depression, and a history of sexual abuse), making diagnosis challenging. Currently, the definitive method to diagnose and stage endometriosis and evaluate the recurrence of disease after treatment is visualization at surgery. Diagnostic laparoscopy is commonly used for the diagnosis.
Long-term treatment of patients with chronic pelvic pain associated with endometriosis involves repeated courses of medical therapy, surgical therapy, or both. In most cases, pain recurs within 6 to 12 months after completion of treatment. Conservative surgical intervention is associated with recurrence of pain. Therefore, medical management is commonly used to alleviate the symptoms of this chronic disorder. There are many randomized placebo-controlled studies showing the superiority of these drugs over placebo (15). As with surgery, there is a significant placebo effect and recurrence of pain after discontinuing the medication is high. Medical suppressive therapy does not influence fertility. The most important principal to remember is that medical management is effective but that symptoms tend to recur rapidly after therapy is stopped. Therefore, first-line medical therapy should focus on drugs that can be used in the long term (16).
Drugs Used for the Treatment of Endometriosis (16):
| Class of Drugs | Drug | Dosage |
| Androgen | Danazol * | 100-400 mg orally twice a day 100 mg per vagina daily |
| Aromatase inhibitor | Anastrozole Letrozole |
1 mg orally daily 2.5 mg orally daily |
| Estrogen - progestin combinations | Monophasic Estrogen-progestin* |
Low ethinyl estradiol dose continuously |
| Gonadotropin - releasing hormone agonist (GnRH) | Goserelin* Leuprolide depot* Nafarelin* |
3.6 mg SC monthly (10.8 mg IM every 3 mo) 3.75 mg IM monthly (11.25 mg IM every 3 mo) 200 mcg intranasally twice a day |
| Gonadotropin - releasing hormone agonist | Cetrorelix | 3 mg SC weekly |
| Progestin | Depo-subQ Provera 104* Dienogest Etonogestrel-releasing implant Levonorgestrel-releasing IUS Medroxyprogesterone acetate Norethindrone acetate* |
104 mg/0.65 mL SC every 3 mo 2 mg daily 1 for 3 years 1 for 5 years 30 mg orally daily for 6 months, then 100 mg IM every 2 weeks for 2 months, then 200 IM monthly for 4 months 5 mg daily |
SC, subcutaneously; IM, intramuscularly; IUS, intrauterine system.
* FDA-approved for endometriosis.
With add-back, i.e., norethindrone acetate 5 mg daily plus vitamin D 800 international units daily plus calcium 1.25 g daily.
Dienogest is a 19-nortestosterone derivative that is approved in the European Union for treatment of endometriosis. It is not available in the United States as a separate drug. It is available only in the oral contraceptive Natazia (Bayer HealthCare Pharmaceuticals; estradiol valerate/dienogest), which is a newer, four-phasic pack that contains dienogest.
Surgical Therapy: Surgical approaches to relieve endometriosis-related pain can be used as first-line therapy or initiated after failed medical therapies (17). Surgical procedures include excision, fulguration, or laser ablation of endometriotic implants on the peritoneum, excision or drainage or ablation of endometriomas, resection of rectovaginal nodules, lysis of adhesions, and interruption of nerve pathways. Randomized, controlled trials have shown that at 6 months, laparoscopic ablation of endometriotic implants is 65% effective in reducing pain, as compared with a 22% rate of pain reduction associated with diagnostic laparoscopy alone (18). A small trial comparing laparoscopic ablation with GnRH agonist treatment showed similar pain reduction with the two approaches (19). Recurrence of pain requiring therapy is common (in 30% to 60% of patients) within 6 to 12 months after treatment (19). An alternative strategy for controlling endometriosis-related pain is interruption of nerve pathways. Whereas ablation of a segment of the uterosacral ligament has not proved effective, randomized, controlled trials have shown the superiority of laparoscopic ablation of endometriotic tissue combined with presacral neurectomy (removal of the nerve bundle within the boundaries of the interiliac triangle) over laparoscopic ablation alone in improving dysmenorrhea and reducing severe midline pain (20). Case stories have shown that hysterectomy with bilateral salpingo-oophorectomy provided pain relief in 80% to 90% of women with debilitating symptoms that were refractory to medical or other surgical interventions; pain was reported to recur in 10% of the women within 1 to 2 years after surgery (21). Post-operative hormone replacement should include both estrogen and a progestogen, since estrogen alone may stimulate growth of microscopic disease.
Adjunctive Medical Therapy: In women with advanced disease (stage III or IV), moderate-to-severe dysmenorrhea, and non-cyclic pelvic pain, post-operative medical therapy may improve pain management by providing control of recurrent microscopic or residual disease. A meta-analysis of six randomized trials that compared 3 to 6 months of postoperative treatment with a gonadotropin-releasing hormone agonist (GnRH), danazol, or combined oral contraceptives with no postoperative treatment of placebo revealed a significant reduction in pain scores at the conclusion of therapy in the active-treatment groups, although the benefits were inconsistent with longer follow-up (to 18 months) after discontinuation of therapy (22). The mean interval between surgery and symptom recurrence requiring alternative therapy was significantly longer for patients who received postoperative treatment with GnRH agonists (>24 months) than for patients who received placebo (12 months) (22).
Management of Endometriosis-Associated Infertility
Even though the causes of endometriosis-associated infertility or subfertility remain obscure, one thing is certain, sub-fertile women have endometriosis at a greater prevalence, 20%-50%, than reproductive-aged women not seeking fertility treatment (23). In one study of infertile women, the incidence distribution was surprisingly robust for minimal-to-mild cases at 68%, whereas women with moderate-to-severe disease constituted the remaining 32% (24). The current classification scheme is not useful in predicting spontaneous pregnancy rates (16). The monthly fecundity rate for those with endometriosis is 0.02-0.10 compared with 0.15-0.20 in fertile couples, although the rate is not necessarily lower than in those patients with unexplained subfertility (16). There is no evidence of an association with recurrent pregnancy loss. Using medical therapy in the form of ovarian suppression for stage I or II endometriosis does not improve fecundity rates and should not be offered (25). Supplementing treatment after operative laparoscopy for stage III or IV endometriosis with GnRH does not appear to be superior to expectant management in terms of natural conception rates during a 5-year follow-up period in a randomized controlled trial (26).
Surgical management of endometriosis-related infertility does improve pregnancy rates, but the magnitude of improvement is unclear. Excision of the endometrioma is more effective than simple drainage and ablation of the cyst wall. Because most endometriomas can be identified by ultrasonography, the number of laparoscopies needed to achieve results is the same. It is important in all surgery for removal of endometriomas that the patient be informed that the surgery may damage the ovary and reduce ovarian reserve (27). The value of excision of deeply infiltrating endometriosis solely to improve pregnancy is unclear and may adverse affect fertility. After initial unsuccessful surgery for endometriosis-associated infertility, in-vitro fertilization (IVF) is the best option rather than reoperation unless pain is still an important issue. Repetitive ovarian surgery has been shown to have a significant negative impact on IVF outcomes (27). Gonadotropin therapy and intrauterine insemination, as well as IVF, are efficacious treatments in women with infertility and endometriosis (28). In a large randomized trial comparing four treatment strategies in 932 couples with stage I or II endometriosis or unexplained infertility, cumulative pregnancy rates during four treatment cycles were as follows: intracervical insemination (10%), intrauterine insemination (18%), gonadotropin therapy and intracervical insemination (19%), and gonadotropin therapy and intrauterine insemination (33%) (16),(29). Ablation of endometriotic lesions with lysis of adhesions is recommended for the treatment of infertility related to stage I or II endometriosis.
Major Guidelines from Professional Societies for the Diagnosis and Management of Endometriosis-Related Pain and Infertility* (7), (21), (28):
| Condition | Recommendation |
| Pain Diagnosis |
Surgery is preferred method for the diagnosis of pelvic pain and a pelvic mass (e.g. endometrioma), but it is not required before initiating empirical therapy, after consideration of other conditions in a differential diagnosis. There should be a low threshold for the evaluation of endometriosis in adolescents because the diagnosis is often missed in this age group. |
| Treatment | Initial treatment is a trial of non-steroidal anti-inflammatory drugs and hormonal therapy (combined oral contraceptives). All hormonal drugs that have been studied (combined oral contraceptives, progestins, GnRH agonists, and danazol) are similarly effective, but their side effects and costs differ. If a GnRH agonist is used, estrogen-progestin add-back therapy is recommended; GnRH agonists are not recommended for adolescents because of their effects on bone. The levonorgestrel intrauterine system is effective in selected patients. Laparoscopic uterosacral nerve ablation is not effective. |
| Infertility Diagnosis |
Both the male and female partner should undergo a full evaluation. |
| Treatment | Super-ovulation with intrauterine insemination provides benefit. Ovarian suppression is not effective in promoting spontaneous pregnancy. The use of GnRH agonist for 3-6 months before IVF and surgical ablation of endometriosis for stage I or II disease are beneficial. Excision of endometriomas >3cm in diameter is of benefit, although there is potential for diminished ovarian reserve. |
* Guidelines are from the American Society for Reproductive Medicine and the European Society of Human Reproduction and Embryology. GnRH denotes gonadotropin-releasing hormone; IVF denotes in-vitro fertilization.
Data on the diagnosis and management of chronic pelvic pain and the treatment of adolescents with pelvic pain are from the American College of Obstetricians and Gynecologists.
Assisted Reproduction and Endometriosis
When treating infertility associated with endometriosis, a stepwise approach is routinely used. Management of endometriosis-associated infertility involves a combination of expectant management, surgery, controlled ovarian stimulation (COS), intrauterine insemination, and advanced assisted reproduction techniques (ART). The use of COS with or without intrauterine insemination may be beneficial for a short course of therapy in patients with endometriosis with tubal patency. If COS does not result in a pregnancy, IVF is a prudent next step. If initial surgery fails to restore fertility in patients with moderate (stage III) or severe (stage IV) endometriosis-related infertility, IVF is an effective alternative. Reproduction for asymptomatic patients offers little added benefit when it comes to infertility (30). Patients with endometriosis appear to have reduced ovarian response to gonadotropins and need a higher dose of gonadotropins for IVF (30). It remains unclear whether the presence or degree of endometriosis is associated with impaired oocytes quality or the fertilization and implantation rate (31). The effect of endometriosis surgery on the outcome of subsequent IVF cycles has been studied, and regression analyses have showed that the time interval between surgery and oocytes retrieval does not affect implantation rates in endometriosis patients undergoing IVF (32).
Endometriosis in Adolescents
Rare cases of endometriosis in premenarchal patients have been described, but the average age at presentation for endometriosis is 15.9 years (33). Approximately 70% of adolescents with pelvic pain not responding to conventional medical therapy have endometriosis (33). Most cases in adolescents can be diagnosed on the basis of history and physical examination. Symptoms suggestive of endometriosis that result in a presumed diagnosis include constant cyclic pain, dysmenorrhea, irregular menses, and deep dyspareunia. The pain is usually severe, disruptive, and interferes with school, sports, and other social activities. Physical examination of an adolescent with endometriosis can vary from a normal examination to generalized pelvic tenderness or persistent adnexal mass. The classic pelvic finding of endometriosis is a fixed retroverted uterus with scarring and tenderness posterior to the uterus. The characteristic nodularity of the uterosacral ligaments and cul-de-sac may be palpated on rectovaginal examination in approximately one-third of women with disease. The ovaries may be enlarged and tender and are often fixed to the broad ligaments or lateral pelvic sidewall. Definitive diagnosis is made by laparoscopic visualization of endometriosis lesions. Adolescents typically have clear, red, white, and yellow-brown lesions more frequently than black or blue lesions.
Medical Management: Non-steroidal anti-inflammatory drugs (NSAIDs) and other analgesics can be used to decrease pain associated with endometriosis. NSAIDs work to limit the production of prostaglandins, which are produced in excess in endometriotic implants. They also function to disrupt the positive feedback mechanism involving local prostaglandin production that is responsible for further increasing estrogen levels in ectopic endometrial cells (34). The result is a decrease in estrogen production, but also a diminished inflammatory response. Oral contraceptive pills (OCPs) are classically first-line treatment in adolescents because of the low side-effect profile and secondary benefit of preventing unwanted pregnancy. OCPs decrease the endometrial lining and thus decrease the amount of tissue that produces prostaglandins. OCPs should be prescribed as a continuous, non-cyclic regimen. This will decrease the number withdrawal bleeds, thus minimizing the occurrence of symptoms. Alternatively, OCPs may induce amenorrhea, resulting in subsequent regression of symptoms. Treatment with OCPs is suppressive but not curative (35).
Progesterone agents include oral or depot medroxyprogesterone acetate, oral 19-nortestosterone derivatives such as norethindrone acetate and norgestrel, and levonorgesterol-releasing intrauterine device. These agents limit estrogen production, cause decidualization of ectopic endometrial tissue, and subsequently result in atrophy of endometrial cells. Danazol decreases volume of ectopic endometrial tissue by inducing hyperandrogenic state. Despite being very efficacious, the androgenic side effects are considerable and irreversible. GnRH agonists create a hypoestrogenic, hypogonadal environment and subsequent down-regulation of gonadotropin secretion resulting in decreased estrogen production. Side effects that limit the use of GnRH agonists include hot flashes, insomnia, decreased bone density. Add-back therapy with norethindrone acetate is recommended to avoid serious long-term sequelae (36). Aromatase mediates the conversion of androstenedione to estrone and testosterone to estradiol (34), (37). Aromatase inhibitors block local estrogen produced by endometriotic tissues, thus destroying the focal feed-back for ectopic endometrial growth. Although there are no randomized controlled trials comparing aromatase inhibitors with traditional medical therapy for endometriosis, pilot studies using aromatase inhibitors with add-back progestins or OCPs have demonstrated significant reduction in endometriosis-associated pelvic pain (37).
Surgical Therapy: Only conservative treatment in which the reproductive organs are conserved is indicated in adolescents. Surgical resection of visible disease can be accomplished by excision, fulguration, and ablation techniques. Depending on the location, depth of invasion, and degree of scarring and inflammation, versatile techniques are applicable. Conservation of reproductive capacity is paramount in the adolescent population. Ablation of endometrial deposits decreases pain, with or without laparoscopic uterine nerve ablation. Presacral neurectomy, a procedure in which the sympathetic nerves from the uterus are divided, may decrease midline abdominal pain. Ablation of ectopic endometrial cells may also increase future fertility in adolescents with endometriosis.
Endometriosis Affecting Non-Reproductive Organs
Extrapelvic endometriosis has been reported in a variety of sites, including the upper abdomen, the diaphragm, the abdominal wall (particularly umbilicus), the perineum (episiotomy scar), and the thorax (38). Remote sites of endometriosis, as in the lung, forearm, thigh, spinal column, and nose have also been reported (39). Perhaps this results from hematologic or lymphatic transplantation of endometrial cells. In fact, 30% of women with endometriosis have disease in the pelvic lymph nodes (39). Actual development of endometriosis likely depends on more than one mechanism of origin. In addition, endometriosis may invade the full thickness of the rectum, large and small bowel, ureters, or bladder. The symptoms that are associated with endometriosis at these sites vary depending on location and depth of infiltration and include women with cyclic episodes of gross hematuria, hemtochezia, and hemoptysis. Although a number of therapeutic approaches have been used for women with presumed extrapelvic endometriosis, the reported efficacy of ovarian suppression with a GnRH agonist appears to support it as the first line of therapy except in cases of obstruction of the ureter or bowel, which are best treated surgically (38). Even in some cases of rectovaginal endometriosis, suppressive medical therapy may alleviate pain (40). Experience with managing these cases is limited to case series, often from a single center.
Long-Term Follow-up
Symptoms may recur in women even after hysterectomy and oophorectomy. Endometriosis may recur in up to 15% of women whether or not the patients are treated with estrogen therapy after bilateral oophorectomy (38). Consequently, hormone therapy with estrogen is not contraindicated after definitive surgery for endometriosis. Although the true rate of recurrence is unknown, in patients with recurrent symptoms undergoing a surgical procedure, endometriotic lesions may be demonstrated. The most common site of recurrent lesions is the large and small bowel (38). These lesions were likely present at the time of surgery and not excised. These lesions can be considered persistent disease rather recurrent disease. Persistence of disease in hypoestrogenic states is most likely due to local expression of aromatase activity. Treatment is difficult and often requires surgery. The use of GnRH agonist in this situation of hypoestrogenemia would not seem to be of benefit. Aromatase inhibitors may be considered but are not uniformly effective (41).
Currently, there are limited data on the timing of estrogen therapy after hysterectomy with bilateral salpingo-oophorectomy for endometriosis. It appears there is no advantage, in terms of recurrence rate, in delaying estrogen treatment after surgery (42). There are limited data to indicate that hormone therapy may stimulate the growth of residual ovarian or endometrial tissue after total hysterectomy and bilateral salpingo-oophorectomy if all visible disease was removed (42). There is also a concern about the possibility of estrogen-induced malignant transformation in residual endometriosis implants. This has led some health care providers to recommend the routine addition of a progestin to the estrogen therapy. However, there is no outcomes-based evidence to support this recommendation. In women with surgical or spontaneous menopause with an intact uterus, progestin therapy is required if estrogen is administered. There appears to be an increased risk of ovarian cancer in endometriosis patients, although it is not clear by what magnitude. It recently has been reported to have a rate ratio of 1.6 (95% confidence interval 1.12-2.09) (43). However, precursors such as cytologic or histologic atypia are uncommon (44).
Summary
Numerous medical treatments are available to treat endometriosis. Current evidence is insufficient to support the superiority of one therapy over another. It is important to consider the patients preference in the treatment approach and to provide appropriate counseling on the risks, side effects, and cost, because endometriosis is a chronic disease that will require long-term treatment. Medical suppressive therapy improves pain symptoms; however, recurrence rates are high after the medication is discontinued. There is significant short-term improvement in pain after conservative surgical treatment; however, as with medical management, there is also a significant rate of pain recurrence. Neither medical nor surgical therapy will permanently alleviate the symptoms associated with endometriosis. Combination therapy involving diagnostic and therapeutic laparoscopy with medical management will likely provide the most optimal results. Various reports show rates of recurrence ranging from 16% to 52%. These differing rates may be in result of variable treatment protocols, different methods of diagnosing recurrence, and inconsistent follow-up. Regular interval evaluations are recommended to address chronic nature of endometriosis. Because of the chronic nature of the disease and the impact on future fertility, psychosocial support is extremely important for this population of women. Surgical management of endometriosis-related infertility does improve pregnancy rates, but the magnitude of improvement is unclear. Excision of an endometrioma is superior to simple drainage and ablation of cyst wall. When relief of pain from treatment with GnRH agonist supports continued therapy, the addition of add-back therapy reduces or eliminates GnRH agonist-induced bone mineral loss and provides symptomatic relief without reducing the efficacy of pain relief. When medical management has failed, undergoing definitive surgical management is appropriate in those who do not desire future fertility.
References
- Giudice LC, Kao LC. Endometriosis. Lancet 2004;364:1789-1799
- Kennedy S, Bergqvist A, Charon C, et al. ESHRE Special Interest Group for Endometriosis and Endometrium Guideline Development Group. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum Reprod 2005;20:2698-2704
- Balasch J, Creus M, Fabregues F, et al. Visible and non-visible endometriosis at laparoscopy in fertile and infertile women and in patients with chronic pelvic pain: a prospective study. Hum Reprod 1996;11:387-391. (Level II-3)
- Bedaiway MA, Falcone T, Mascha EJ, et al. Genetic polymorphism in the fibrinolytic system and endometriosis. Obstet Gynecol 2006;108:162-168. (Level II-2)
- Giudice LC, Swiersz LM, Burney RO. Endometriosis. In: Jameson JL, DeGroot LJ, eds. Endocrinology. 6th ed. New York: Elsevier, 2010:2356-2370
- Bulun SE. Endometriosis. N Engl J Med 2009;360:268-279
- Giudice LC. Endometriosis. N Eng J Med 2010;362(25):2389-2398
- Lebovic DI, Mueller MD, Taylor RN. Immunobiology of endometriosis. Fertil Steril 2001;75:1-10
- Stratton P, Berkley KJ. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update 2011;17:327-346
- Lemons NA, Arbo E, Scalco R, et al. Decreased anti-Müllerian hormone and altered ovarian follicular cohort in infertile patients with mild/minimal endometriosis. Fertil Steril 2008;89:1064-1068
- Mansour G, Aziz N, Sharma R, et al. The impact of peritoneal fluid from healthy women and from women with endometriosis on sperm DNA and its relationship to the sperm deformity index. Fertil Steril 2009;92:61-67
- Walter AJ, Hentz JG, Magtibay PM, et al. Endometriosis: correlation between histologic and visual findings at laparoscopy. Am J Obstet Gynecol 2001;184:1407-1411; discussion 1411-1413. (Level II-3)
- Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril 1997;67:817-821. (Level III)
- ACOG Practice Bulletin no. 51: chronic pelvic pain. Obstet Gynecol 2004;103:589-605
- Vercellini P, Crosignani P, Somigliana E, et al. Waiting for Godot: a commonsense approach to the medical treatment of endometriosis. Hum Reprod 2011;26:3-13
- Falcone T, Lebovic DI. Clinical management of endometriosis. Obstet Gynecol 2011;118:691-705
- Jacobson TZ, Duffy JM, Barlow D, et al. Laparoscopic surgery for pelvic pain associated with endometriosis. Cochrane Database Syst Rev 2009;4:CD001300
- Berkley KJ, Rapkin AJ, Papka RE. The pains of endometriosis. Science 2005;308:1587-1589
- Prentice A, Deary AJ, Goldbeck-Wood S, et al. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst Rev 2000;2:CD000346
- Proctor ML, Latthe PM, Farquhar CM, et al. Surgical interruption of pelvic nerve pathways for primary and secondary dysmenorrhea. Cochrane Database Syst Rev 2005;4:CD001896
- Idem. Treatment of pelvic pain associated with endometriosis. Fertil Steril 2008;90:Suppl:S260-S269
- Yap C, Funress S, Farquhar C. Pre- and post-operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev 2004;3:CD003678
- Ozkan S, Murk W, Arici A. Endometriosis and infertility: epidemiology and evidence-based treatment. Ann N Y Acad Sci 2008;1127:92-100
- DHooghe TM, Debrock S, Hill JA, Meuleman C. Endometriosis and subfertility: is the relationship resolved? Semin Reprod Med 2003;21:243-254
- Hughes E, Brown J, Collins JJ, et al. Ovulation suppression for endometriosis. Cochrane Database Syst Rev 2007;3:CD000155
- Loverro G, Carriero C, Rossi AC, et al. A randomized study comparing triptorelin or expectant management following conservative laparoscopic surgery for symptomatic stage II-IV endometriosis. Euro J Obstet Gynecol Biol 2008;136:194-198
- Busacca M, Vignali M. Endometrioma excision and ovarian reserve: a dangerous relation. J Minim Invasive Gynecol 2009;16:142-148. (Level III)
- Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility. Fertil Steril 2006;86:Suppl:S156-S160
- Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril 2002;77:1148-1155
- Mahutte NG, Arici A. Endometriosis and assisted reproductive technologies: are outcomes affected? Curr Opin Obstet Gynecol 2001;13:275-279
- Bedaiwy M, Shahin AY, AbulHassan AM, et al. Differential expression of follicular fluid cytokines: relationship to subsequent pregnancy in IVF cycles. Reprod Biomed Online 2007;15:321-325
- Bedaiwy MA, Falcone T, Katz E, et al. Association between time from endometriosis surgery and outcome of in vitro fertilization cycles. J Reprod Med 2008;53:161-165
- Laufer MR, Sanfilippo J, Rose G. Adolescent endometriosis: diagnosis and treatment approaches. J Pediatr Adolesc Gynecol 2003;163(3 Suppl):S3-S11
- Hartel Z. Dysmenorrhea in adolescents and young adults: etiology and management. J Pediatr Adolesc Gynecol 2006;19(6):363-371
- Doyle JO, Missmer SA, Laufer MR. The effect of combined surgical-medical intervention on the progression of endometriosis in an adolescent and young adult population. J Pediatr Adolesc Gynecol 2009;22(4):257-263
- Vercellini P, Fedele L, Pietropaolo G, et al. Progestogens for endometriosis: forward to the past. Hum Reprod Update 2003(9(4):387-396
- ACOG. ACOG Committee Opinion: Aromatase inhibitors in gynecologic practice. Obstet Gynecol 2008;112(2 Part 1):405-407
- ACOG Practice Bulletin. Management of endometriosis. Number 114, July 2010. Obstet Gynecol 2010;116:223-236
- Laufer MR. Current approaches to optimizing the treatment of endometriosis in adolescents. Gynecol Obstet Invest 2008;66 (Suppl 1):19-27
- Vercellini P, Somigliana E, Vigano P, et al. Surgery for endometriosis-associated infertility: a pragmatic approach. Hum Reprod 2009;24:254-269. (Level III)
- Bohrer J, Chen CC, Falcone T. Persistent bilateral ureteral obstruction secondary to endometriosis despite treatment with an aromatase inhibitor. Fertil Steril 2008;90:2004.e7,2004.e9. (Level III)
- Matorras R, Elorriaga MA, Pijoan JI, et al. Recurrence of endometriosis in women with bilateral adnexectomy (with or without total hysterectomy) who received hormone replacement therapy. Fertil Steril 2002;77:303-308. (Level I)
- Aris A. Endometriosis-associated ovarian cancer: a ten year cohort study of women living in the Estric Region of Quebec, Canada. J Ovarian Res 2010;3:2
- Bedaiwy MA, Hussein MR, Bicotti C, Falcone T. Pelvic endometriosis is rarely associated with ovarian borderline tumors, cytologic and architectural atypia: a clnicopathologic study. Pathol Oncol Res 2009;15:81-88
Published: 27 June 2012
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


