Robotic Gynecologic Surgery
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers.
Educational grant provided by Women's Health and Education Center (WHEC).
Minimally invasive surgical techniques for performing hysterectomies and other gynecologic surgeries have been shown to reduce patient morbidity and shorten hospital stay. Laparoscopy has become a forerunner in the pursuit of improving surgical outcomes by reducing postoperative pain and decreasing recovery time. However, the use of laparoscopic instruments can be cumbersome and unwieldy, thereby increasing operative time and reducing dexterity, which can limit the surgeon's ability to perform the procedure with the same techniques that can be accomplished through a traditional abdominal incision. Introduced in 1999, the daVinci Surgical System, the urologists are still considered the system's number one user, but robotic applications in gynecologic surgery have been expanding. In 2005, U.S. Food and Drug Administration approval was obtained for use of the daVinci robot (Intuitive Surgical, Inc., Sunnyvale, CA) for use in gynecologic surgery as a modification of the laparoscopic approach. This robot employs the use of robotic arms that control modified laparoscopic instruments that have seven degrees of freedom, giving the surgeon significantly improved dexterity. The advent of this technology has made it possible to perform the traditional gynecological procedures through a laparoscopic technique that allows for ease of maneuvering, thus combining the benefits of a minimally invasive surgical procedure with a shorter recovery period. The use of robotics in gynecologic surgery is increasing in the United States. A decade later, growing numbers of health-care institutions are purchasing the robotic system. In gynecology the expansion is reflected in literature reports on robotic applications in general gynecology, urogynecology/pelvic reconstructive surgery, gynecologic oncology, and reproductive endocrinology.
The purpose of this document is to review the recent adoption, experience, and applications of robot-assisted laparoscopy in gynecologic surgery. It will likely continue to develop as more gynecologic surgeons are trained and more patients seek minimally invasive surgical options. Well-designed, prospective studies with well-defined clinical, long-term outcomes, including complications, cost, pain, return to normal activity, and quality of life, are needed to fully assess the value of this new technology. Surgical innovation is necessary. There are ethical and societal issues that remain incompletely understood about the use of robotic surgery.
History and Development of Robot-assisted Surgery:
The use of robots in surgery has only come about within the past 25 years. The first application of a robot surgery was in neurosurgery. The limitations encountered with conventional laparoscopy include counter-intuitive hand movements, two dimensional visualization, and limited degrees of instrument motion within the body as well as ergonomic difficulty and tremor amplification (1). In an attempt to overcome these obstacles, robotics has been recently incorporated in gynecologic armamentarium. The original model, known as the PUMA 560 (Stäubli Corporation, Duncan, SC), was used for neurosurgical stereotactic maneuvers under computed tomography (CT) guidance. Soon, orthopedic surgery was using a device called ROBODOC (Curexo Technology Corporation, Sacramento, CA) to aide in total hip replacements, and urology was performing transurethral resection of the prostate with a robot through guidance from a preoperatively constructed three-dimensional image. Common among these early robots was the fact that they were developed to function autonomously with a preoperative plan or in a supervisory role. This passive role would evolve into a more active one, with an immersive environment that became known as robotic telepresence technology. The concept of robotic telepresence technology was born through the collaborative efforts of the Stanford Research Institute, the Department of Defense, and the National Aeronautics and Space Administration (NASA) (2). The research was directed toward allowing military surgeons to perform surgery on wounded soldiers from a safe and remote location. As the technology further developed, research focused not on telesurgery but on using robotic surgery to further enhance laparoscopic and minimally invasive surgery in civilian operating rooms. Such advances became possible due to improved vision systems, instruments with articulating distal ends, and improved ergonomics (1)(2).
Although robotic telepresence technology was initially created for cardiac surgery, it soon was applied to the fields of urology and gynecology. Today there is only one U. S. Food and Drug Administration approved device for surgical robotics. This current robotic platform is known as the daVinci surgical system (Intuitive Surgical, Sunnyvale, CA). The key technologic advancements seen with earlier predecessors are not only incorporated into today's platform but are further refined. In 2000, the US Food and Drug Administration approved the use of a robotic system for laparoscopy, and specifically for hysterectomy in 2005. Robotic surgery allows a surgeon to sit at a console while three or four robotic arms move over the patient according to the surgeon's commands. Commonly performed robot-assisted laparoscopic surgeries in gynecology include benign hysterectomy, myomectomy, tubal reanastomosis, radical hysterectomy, lymph node dissections, and sacrocolpopexies.
Indications and Contraindications for Robot-assisted Surgery:
Robot-assisted surgery has similar indications as conventional laparoscopic surgery. However, because robotic technology allows surgeons to more easily and simply perform complex laparoscopic maneuvers, the availability of the robot would allow the surgeon to perform more sophisticated procedures than the conventional laparoscopic route, therefore reducing the number of open procedures. For example, a gynecologic surgeon might ordinarily prefer an open procedure over the conventional laparoscopic route for advanced pelvic endometriosis. However, with the availability of the robotic system, which facilitates tissue dissection and suturing, the surgeon may elect the robotic approach, resulting in reduced morbidity, shorter hospitalization, less post-operative pain, earlier return to work, and better cosmetic results compared to an open operation. Contraindications, too, are usually similar to those of conventional laparoscopy (e.g., immediate need for laparotomy to control bleeding, poor visualization, or exposure). The robotic system usually shortens the operative time of a long endoscopic procedure (e.g., radical hysterectomy, lymphadenectomy, sacrocolpopexy). On the other hand, it may increase the total time of a short simple procedure (e.g., adnexectomy, endoscopic sterilization). When you add in the time spent in draping and setting up the robot, docking time, and operating room turnover time, using the robotic system in simple short endoscopic procedures may not be cost effective.
Basic Set-up and Instruments:
A basic surgical robotic system is composed of three parts: a patient-side robot, a vision cart, and the robotic master console. The robotic surgeon operates from the remote master console using a combination of hand controls and foot pedals. One foot pedal controls the camera movement (right/left, up/down, in/out) and horizontal orientation, while a nearby pedal controls the focus. Another pedal provides a clutching mechanism that allows for repositioning of hand controls and provides the instruments a range of motion beyond the physical confines of the console. Another set of pedals controls both monopolar and bipolar energy sources. The patient-side cart is wheeled in between the patient's legs, and the robotic arms are attached to stainless steel robotic trocars through a process termed "docking". The hand controls operate either the camera or up to two robotic instruments at one time. There are up to three operative robotic arms, with the option to swap control among any two of the three operative arms. While operating the robotic operative instruments, the surgeon is capable of manipulating, repositioning, grasping, retracting, cutting, dissecting, coagulating, and suturing. The robotic master console also provides the surgeon with three-dimensional imaging through a stereoscopic viewer. The following instruments are used for most of the gynecologic surgeries:
n
daVinci Robot and EndoWrist® Instruments commonly used for gynecologic surgeries.
Unlike industrial robots, these systems are not autonomous, and to be taxonomically correct, they ought not to be called machines. A surgical robot is actually a collection of wristed tools called manipulators, which receive digital instructions from an interfaced computer. The surgeon, seated at an ergonomically designed video console with an "immersive" 3-D display, initiates the digital instructions by controlling sophisticated hand grips -- essentially, joysticks with seven degrees of freedom, (adding the pitch, the yaw and the pincer-like movement to those that were already available). The manipulators inside the patient's body duplicate the surgeon's hand movements at the console, and software filters out even physiologic hand tremors (3). Despite all of these technologic advancements that make the surgeon nearly autonomous, a bedside assistant is still required for all robot-assisted cases. Their responsibility is mainly instrument exchanges, suction and irrigation, suture introduction and retrieval, and additional retraction.
n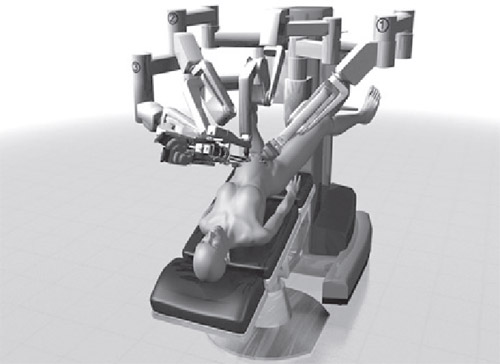
Patient postioning; setting up and docking the robot for robotic-assisted gynecologic surgeries.
Docking the Robot and Instrument Selection:
The system's two main components are the surgeon's console and the robotic column. After endotracheal intubation, position the patient in the dorsal lithotomy position with both arms tucked comfortably. A 12-mm umbilical trocar is placed using the open Hasson technique. Two 8-mm specially designed robotic trocars are placed bilaterally 10 cm lateral to and at the level of the umbilicus. Meanwhile, an accessory 10-mm trocar is placed between the umbilical port and the left lateral port 3 cm cephalad to the umbilicus. When the fourth robotic arm is used, that trocar is placed 10-cm lateral and 10 cm caudal to the right robotic trocar. The operating room table is lowered to the lowest level and enough Trendelenburg is obtained to permit removing the bowel out of pelvis, up to 30º in obese patients. The robotic column is advanced and placed between the patient's legs. Next, the robotic laparoscope (InSite Vision System; Intutive Surgical) is attached to the umbilical trocar, and the two robotic arms are attached to the lateral robotic trocars. The surgeon sits, unscrubbed, at the console. The assistant sits on the patient's left side using the right hand to assist the surgeon through the accessory port (e.g., retraction, introduction of sutures and suction irrigation, vessel sealing, and specimen retrieval) and vice versa for a left-handed assistant. With S and Si platform side docking makes vaginal access easier.
n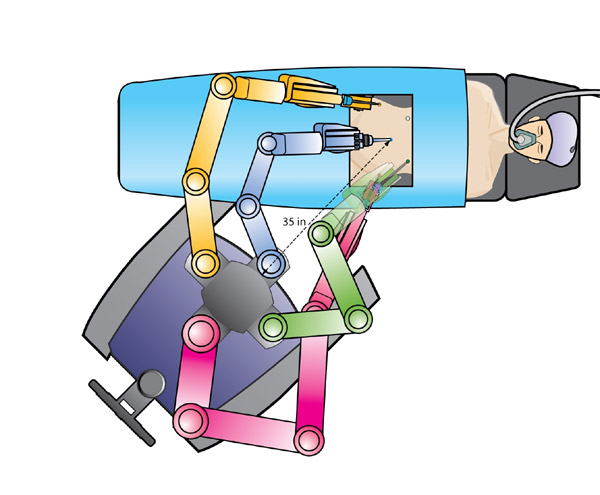
S and Si Platform set up for side docking
A variety of EndoWrist® instruments are available for robotic gencologic surgery; however, the surgeon should limit intrument exchange for an efficient procedure and to minimize cost. For most patients the monopolar spatula or scissors are used with the robotic right arm, and PK (plasma kinetic) on the robotic left arm. A grasper (Prograsp) instrument is inserted with the robotic fourth arm whenever it is used. When suturing is required, the robotic right instrument is switched for a Mega needle holder, which is also capable of cutting sutures.
Advantages of Robotic Surgery:
Robotic surgery offers three advantages over laparoscopy: a three-dimensional vision system, wristed instrumentation, and ergonomic positioning for the surgeon while performing surgical procedures. Conventional laparoscopic surgery has a steep learning curve for physicians because it has two-dimensional imaging and involves mastering counter-intuitive hand movements. Robotic surgery, however, solves some of these challenges because it uses three-dimensional imaging and instruments that move just like the surgeon's wrist. The robotic system eliminates normal hand tremors and allows the surgeon to sit, instead of stand, during the procedure, which helps guard against fatigue. The gynecologic laparoscopic surgeon performs procedures in a confined space, the female pelvis. Wristed instrumentation allows the gynecologic surgeon to obtain the exact instrument angle available at laparotomy. This also eliminates the fulcrum effect that is present with laparoscopy, where surgeons need to move their hand in the opposite direction to the intended location of the distal instrument tip (eg, toward the patient's left if they want the instrument moved to the patient's right). With robotic surgery, the movements are natural, and surgeon moves their hands in whichever direction they want the instruments to move. The "wristed" instrumentation affords greater dexterity and provides seven degrees of freedom, similar to the human hand (4). Three degrees are provided by the robotic arms attached to the abdominal wall trocars (insertion, pitch, yaw), and four degrees result from the "wristed" instruments (pitch, yaw, roll and grip). The terms pitch, roll and yaw are the three characteristics that describe the rotations in three dimensions around the robotic instrument's coordinate system origin, the center of mass. Pitch is the rotation around the lateral axis. The yaw is rotation about the vertical axis, and the roll is rotation around the longitudinal axis.
Decreased blood loss has been reported in comparative studies (5). The enhanced visualization gives the gynecologic surgeon an improved ability to identify tissue planes, blood vessels, and nerves while performing the surgical procedure. Fatigue and physical discomfort can become limitations during any surgical procedure. With robotic surgery, the surgeon sits comfortably at the surgical console from the vantage point of standing at the patient's head and manipulates the hand controls and foot pedals while in an ergonomic position. This may serve to reduce fatigue and discomfort during complex surgical procedures. TilePro (Intuitive Surgical, Inc. Sunnyvale, CA) is a feature that allows for image and video input to the console. This can be viewed by the console surgeon for instructional purposes during the early phase of the learning curve or for input of radiologic data from ultrasonography, computed tomography (CT), or magnetic resonance imaging (MRI).
Disadvantages of Robotic Surgery:
The daVinci robot costs $ 1.65 million to purchase with annual maintenance costs of this technology is $ 149,000 per year. Minimally invasive hysterectomy approaches (vaginal and laparoscopic) are underused in the United States. For example, of hysterectomies done for the top seven non-cancer diagnoses in the United States, approximately 66.1% were abdominal, 21.8% were performed vaginally, and only 11.8% were performed laparoscopically (4)(6). Will robot surgery substantially improve outcomes over vaginal or conventional laparoscopic routes? Introduced in the early 1990s, laparoscopic-assisted vaginal hysterectomy (LAVH) was expected to replace the "blind" vaginal hysterectomy; yet in a randomized trial, the only significant difference between these surgical approaches was twice the cost to include the laparoscope. Follow-up studies did show advantages to the LAVH relative to hysterectomy done by laparotomy, yet these numbers have been stable while vaginal hysterectomy numbers have declined, presumably at the expense of LAVH. Considering these facts, many researchers believe that laparoscopic hysterectomy as it is currently practiced has contributed to no substantial improvement in patient care but has lead to increased health care costs (6)(7). Moreover, increased hospital market share evaporates as more hospitals acquire the daVinci robot -- in some cases without yet identifying a surgical team that has intention to use it.
The main disadvantages of robotic surgery across applications are the cost, the large size of the robot console, limited availability within some health systems, lack of tactile feedback or haptics, and the need to train residents, attending surgeons and operating room personnel on the use of this technology. Additional costs that need to be considered include the time and cost of training surgeons and operating room personnel, the potential cost or reduced productivity during a surgeon's learning curve, and increased operative time associated with operating room setup as well as the assembly and disassembly of the robotic system during the early phase of adoption. There is evidence that with experience, operative time can become shorter with laparoscopy (5). Docking time has been shown to decrease with experience. Once docked, the robotic arms are attached and fixed to specialized trocars. Because the operating table and the robot do not communicate and are therefore not synchronized, once the robotic unit is docked, the patient bed cannot be moved in any direction, including Trendelenburg; otherwise, the trocar depth can become incorrectly positioned and abdominal wall as well as visceral trauma could occur.
Vaginal cuff dehiscence with small bowel evisceration after hysterectomy is a rare event that may be occurring more frequently with the advent of robotic laparoscopic hysterectomies. Only 59 cases were reported between January 1900 and December 2001. 95% of cases performed occurred after total abdominal (TAH) or vaginal hysterectomy (TVH). Recent reviews describe a disproportionate number of vaginal cuff dehiscences occurring after total laparoscopic hysterectomy (TLH), suggesting that vaginal cuff dehiscence may be more common complication of laparoscopic hysterectomy than TAH or TVH. A retrospective review of 2,399 hysterectomies performed at the Mayo Clinic in Scottsdale, Arizona, of which 15% were performed robotically and 9% with traditional laparoscopy reported the vaginal cuff dehiscence rate to be 2.87% for robotic hysterectomy and 0.47%, 0.13% and 0.99% for TLH, TVH, and TAH respectively. Colpotomy technique may account for the observed increased risk of vaginal cuff dehiscence associated with robotic surgery. This observation may be because of thermal spread and cuff tissue damage from electro-surgery used for colpotomy (8). Monopolar hook and a blend of coagulation and cutting modes theoretically can reduce the risk of vaginal cuff dehiscence. Alternatively, a harmonic scalpel which uses a high frequency vibrating blade for cutting may be used during both robotic and traditional TLH.
Applications of Robot-assisted Surgery in General Gynecologic:
Robotics has been looked upon as a possible way to facilitate the trend toward a less invasive hysterectomy and patient outcomes. The availability of the robotic system should not dictate the route of hysterectomy (endoscopic vs. vaginal). Whenever technically feasible and medically appropriate, patients requiring hysterectomy should be offered the vaginal approach because morbidity appears to be lower with vaginal approach than with any other method. Endoscopic hysterectomy is indicated in the following cases: lysis of adhesions; treatment of endometriosis; management of uterine leiomyomata, and/or adnexal masses that complicate the performance of vaginal hysterectomy; ligation of infundibulopelvic ligaments to facilitate difficult ovary removal; and evaluation of the pelvic and abdominal cavity before hysterectomy. In 2002 this study (9) reported one of the earliest experiences with robot-assisted laparoscopic hysterectomy. This series include 16 patients ranging in age from 27 to 77 years. Operative time ranged from 270 to 600 minutes, and blood loss ranged between 50 mL and 1,500 mL, with an average loss of 300 mL. The average stay was 2 days, with a range of 1 to 3 days. Although their approach was labeled a laparoscopic hysterectomy, all cases in that series were Type IIB according to the American Association of Gynecologic Laparoscopists (AAGL) classification system of laparoscopic hysterectomy, meaning that the posterior culdotomy and ligation of the cardinal and uterosacral ligament complexes were performed vaginally to complete the hysterectomy (9).
The comparative study (5) to date of robotic hysterectomy to conventional laparoscopy is a retrospective review of 200 consecutive hysterectomy cases completed before and after implementation of a robotics program. There were no statistically significances in patient characteristics or uterine weights between the two groups. The rate of intraoperative conversion to laparotomy was two-fold higher in the laparoscopic cohort of 100 patients as compared with the robotic cohort (9% compared with 4%). The mean blood loss was also significantly reduced in the robotic cohort. However, the incidence of adverse events was similar in the two groups. Mayo Clinic, Scottsdale, published the largest series to date (11); ninety-one patients undergoing robotic hysterectomy were evaluated. Those requesting lymphadenectomy were excluded. A wide range of pathology, including ovarian neoplasms, abnormal uterine bleeding, and moderate-to-severe endometriosis, was addressed. The average uterine weight was 135.5 grams. The mean operative time was 127.8 minutes, with an estimated blood loss of only 78.6 mL and hospital stay of 1.4 days. There were no conversions to conventional laparoscopy or laparotomy and no bladder or ureteral injuries occurred.
n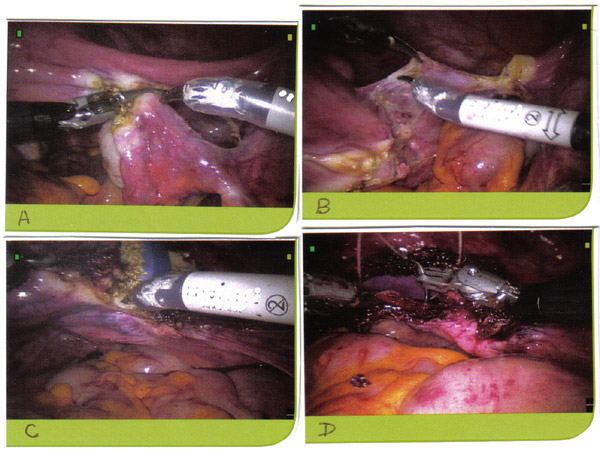
Robot-assisted hysterectomy for benign conditions; A. Skeletonizing and coagulating vascular pedicles; B. Vesico-uterine reflection; C. Colpotomy; D. Vaginal cuff closure.
Applications of Robot-assisted Surgery in Gynecologic Oncology:
A natural progression of robotic technology in gynecology has been to the area of oncology. Early experiences clearly demonstrated the feasibility of applying robotic assistance to laparoscopic cancer staging without an increase in complication rates or compromise to surgical technique. There were no statistically significant differences between these three approaches: laparotomy, conventional laparoscopic, and robotic, with respect to mean age, body mass index, or lymph node count (13). However, the authors did find significantly less estimated blood loss and shorter length of stay associated with the robotic approach. In this study, operative times were comparable to open surgery and better than conventional laparoscopy. There were also no conversion or intraoperative complications in the robotic group (12). Similarly Boggess et al published a study comparing robot-assisted, conventional laparoscopic and open hysterectomy with staging for endometrial cancer (14). They found the highest lymph node yields with the robotic approach. Robotic hysterectomy with staging was associated with significantly longer operative time, in this series, as compared with open hysterectomy, but shorter operative times when compared with laparoscopic group. Ultimately, 5-year survival rates will need to be evaluated to truly assess the effect of robotics on gynecologic cancer staging. Robotic surgery is a useful minimally invasive tool for the comprehensive surgical staging of the obese and morbidly obese women with endometrial cancer (12)(14). As this patient population is at increased risk of death from all causes, including post-operative complications, all efforts should be made to improve their outcomes and minimally invasive surgery provides a useful platform by which this may occur.
Applications of Robot-assisted Surgery in Reproductive Endocrinology:
Robotic technology also facilitates the dissection of the myoma and suturing of the uterine incision. In a retrospective case-matched study comparing robotic myomectomy to open myomectomy, investigators reported longer operative times in the robotic group [mean: 231 (+/-85) minutes vs. 154 (+/- 43 minutes, P<.05], but decreased blood loss [mean: 195 (+/- 228) mL vs. mean 364 (+/- 473 mL, P<.05] and shorter length of stay [mean: 1.4 (+/-0.9 days vs. 3.62 (+/- 1.5 days, P<.05] when compared with the laparotomy group (15). Because various steps of myomectomy can be difficult with conventional laparoscopy, many concerns exist. In particular, the ability to enucleate leiomyomas and perform a multilayer closure requires advanced laparoscopic skills. Although pregnancy rates after myomectomy managed endoscopically are similar to those after laparotomy, a major worry continues to be the risk of uterine rupture. Also, the risk of recurrence seems to be higher after laparoscopic myomectomy compared with laparotomy. These factors and the associated learning curve may contribute to the fact that abdominal myomectomy remains the standard approach (16).
n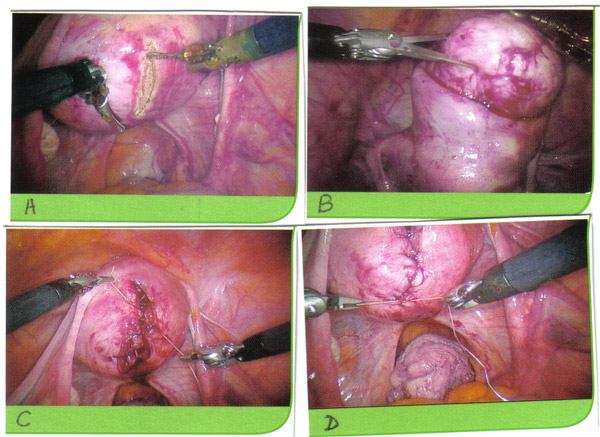
Robot-assisted myomectomy; A. Hysterotomy; B. Enucleation; C. Multi-layered suture closure of defect -- deep layers; D. Multi-layered suture closure of defect -- superficial layer.
The ability to leverage the advantages of an advanced vision system along with the microsurgical precision of articulating endoscopic instruments is exemplified in gynecologic surgery with tubal reanastomosis. In 2007, Dharia et al (4) prospectively compared robotic-assisted tubal anastomosis to open microsurgical tubal anastomosis in women with history of bilateral tubal ligation who desired reversal. They found robotic tubal anastomosis was associated with significantly longer operative time (201 minutes compared with 155 minutes with open) but significantly shorter hospital length of stay (4 hours compared with 34.7 hours with open) and faster return to normal activities of daily living (11.1 days compared with 28.1 days with open). Pregnancy rates were comparable between groups (62.5% compared with 50% with open), yet the robotic group had a higher number of ectopic pregnancies (4 compared to 1 with open). The cost per delivery was similar between robotic anastomosis ($92,488) and open tubal anastomosis ($92,206).
Applications of Robot-assisted Surgery in Urogynecology/Pelvic Reconstruction:
Sacrocolpopexy is probably the fastest growing reconstructive procedure performed robotically. The procedure involves extensive dissection of presacral space and rectovaginal septum, mesh positioning, suturing, and extra-corporal suturing and knot tying, which is simplified by the robotic system. There are four main studies on robotic sacrocolpopexy. The major strength of this retrospective cohort study (17) is the fact that authors report on outcomes of robotic sacrocolpopexy in comparison with a control group of patients who underwent abdominal sacrocolpopexy, the criterion standard for advanced vaginal vault prolapse. It is critical to include a control group for comparison to assess the efficacy of this newer minimally invasive surgical technique. It is worth noting that the control group in this study comprised patients at the same institution, exposed to the same practice patterns regarding perioperative care, including similar surgical technique, antibiotic prophylaxis, and postoperative discharge guidelines as the robotic group. Another strength of this study is the use of objective data, in the form of POP-Q scores, for outcome assessment. This minimizes bias that may be introduced by the retrospective design of the study. Robotic sacrocolpopexy demonstrated similar short-term vaginal vault support compared with abdominal sacrocolpopexy, with less blood loss and shorter length of stay. Operative time was longer but may decrease as the learning curve for this new procedure improves. There were similar outcomes between the two groups in terms of perioperative complications, but this is limited by the low incidence of these complications. Long-term data are needed to assess the durability of this newer minimally invasive approach to prolapse repair. Robotic vesicovaginal and ureterovaginal fistula repair has been reported through small case series and case reports. In a case series of seven patients with vesicovaginal fistula, the researchers reported an average operative time of 141 minutes (range 110 to 160). Mean blood loss was 90 mL, and no significant intraoperative or postoperative complications were observed (18).
Learning Curve and Credentialing:
The belief is that robotic surgery will allow for a more rapid development of the necessary skills and allow for a larger number of surgeons to attain those skills, and therefore, provide minimally invasive surgical options to a larger number of patients. There is a paucity of experienced robotic surgeons. Currently, the training involves practice with the surgical robot in either a pig or human fresh tissue environment to become familiar with the functions of the robot, the attachment of the robotic arms to the robotic trocars, and the overall functions of the robotic console. Further training allows surgeon to learn how to perform simple maneuvers such as grasping, cutting, and intracorporeal knot tying, the last task being particularly difficult with conventional laparoscopy. Two studies have specifically looked at learning curves. A recent study (19) evaluated 113 sequential patients over a 22-month period. They found that the learning curve for various benign surgical interventions stabilized in regard to operative time times after 50 cases. A similar learning curve was documented for the operating room team to be able to set up the robot for surgery in 30 minutes. This break point was 20 cases. A major hurdle often encountered early in a surgeon's robotic experience is "docking time", or the attachment of the robotic device to the patient.
With the introduction of robotic surgery, hospitals and departments have been challenged to establish credentialing requirements for this advanced surgical technique. There are no universally established credentialing guidelines. Most healthcare facilities require performing a minimum of two robotic surgical procedures of each type for which privileges are being requested in the presence of an expert preceptor. Some institutions are using four as the minimum number of proctored robotic surgeries necessary for independent robotic privileges. An expert preceptor is defined as a surgeon who has current Robotic Surgical Privileges and has been approved as an expert preceptor by the Chair of the Department of the individual applying for privileges. Similar to other surgical procedures maintenance of competence requires performing procedures on an ongoing basis.
Future of Robotic Surgery:
In this high-tech future, surgery could be performed skillfully and promptly even in dangerous or inaccible places. In addition to small or narrow places in the human body, remote places in the world are often mentioned by those invoking the promise of robotic surgery. The logical extension of this mode of operation -- telesurgery might be a good way to distribute health care in the developing countries. And NASA is exploring its potential for the proposed manned mission to the Moon and to Mars. Many of the disadvantages could be improved with further development and technologic refinement. Future research is imperative to address the questions of cost-effectiveness, effect on resident training, and whether this technology is best made available to all surgeons or to a limited number of surgeons with high surgical volume who develop particular robotic expertise and are able to maintain proficiency with this evolving technology.
Telestration, the ability to write on a touch screen and have the markings visible at the console, is helpful but does not replace the educational guidance a "co-sugeon" training robotic console would afford. Such a "co-pilot" console is currently commercialy available with Si platform. Even as the current systems begin to be put in practice, the true visionaries are focusing on the surgical robot less as a mechanical device than as an information system -- one that should be fused with other information systems. One proposed example of this kind of fusion is image-guided surgery, also called surgical navigation. Robot-assisted surgeons will be able to see real-time, three-dimensional scanner images electronically superimposed over the operative field that is displayed on the monitor (20). In other words, on the screen, human anatomy will be rendered translucent, and the surgeon will be able to determine the exact location of a tumor and more readily avoid damaging vital structures -- such as the major intraparenchymal vessels and bile ducts that are sometimes accidentally severed or the inferior vana cava during lymph-node dissection during gynecologic oncology cases or during right adrenalectomy. In fact, with preoperative scanner images, surgeons could robotically practice their patients' surgery the night before, and the robot's computer could be programmed not to allow its instruments to penetrate the vena cava, thereby eliminating bloody intraoperative mishaps.
Summary:
Robotic surgery has seen enormous growth over the past decade. Because robotic technology allows surgeons to easily and simply perform complex laparoscopic maneuvers, it has already revolutionalized laparoscopic urologic and oncologic surgery. Several studies done have linked robotic technology with faster performance, better accuracy, faster suturing, and fewer errors when compared to conventional laparoscopic intrumentation. Advantages include: dexterity, precision, three dimensional imaging and reduced surgeons's fatigue. Its limitations are: lack of tactile feedback, increased cost, a bulky system. Well-designed randomized trials comparing routes of surgery with clinically meaningful long-term outcomes are needed. These outcomes include effect on quality of life and patient satifaction associated with hysterectomy, recurrence, and survival rates for oncology procedures, future fertility rates and pregnancy outcomes associated with tubal reanastomosis or myomectomy, and reoperation rates for urinary incontinence or recurrent pelvic organ prolapse.
Acknowledgement:
Gratitude is expressed to Dr. James L. Whiteside, Department of Obstetrics and Gynecology, Dartmouth Medical School and Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire (USA) for the expert opinions and reviewing the paper. Special thanks to Kendra Martell and Jonathan Conta, Intuitive Surgical Inc., Sunnyvale, CA for the assistance with the research and support.
References:
- Stylopoulos N, Rattner D. Robotics and ergonomics. Surg Clin North Am 2003;83:1321-1337
- Satava RM. Robotic surgery: from past to future -- a personal journey. Surg Clin North Am 2003;83:1491-1500,xii
- Camarillo DB, Krummel TM, Salisbury JK Jr. Robotic surgery: past, present, and future. Am J Surg 2004;188:Suppl:2S-15S
- Visco AG, Advincula AP. Robotic gynecologic surgery. Obstet Gynecol 2008;112:1369-1384
- Payne TN, Dauterive FR. A comparison of total laparoscopic hysterectomy to robotically assisted hysterectomy: surgical outcomes in a community practice. J Minim Invasive Gynecol 2008;15:286-291
- Wu JM, Wechter ME, Geller EJ et al. Hysterectomy rates in the United States, 2003 Obstet Gynecol 2007;110:1091-1095
- Meeks GR. Advanced laparoscopic gynecologic surgery. Surg Clin North Am 2000;80:1443-1464
- Robinson BL, Liao JB, Adams SF et al. Vaginal cuff dehiscence after robotic total laparoscopic hysterectomy. Obstet Gynecol 2009;114:369-371
- Diaz-Arrastia C, Jurnalov C, Gomez G et al. Laparoscopic hysterectomy using a computer-enhanced surgical robot. Surg Endosc 2002;16:1271-1273
- Olive DL, Parker WH, Cooper JM et al. The AAGL classification system for laparoscopic hysterectomy. Classification committee of the American Association of Gynecologic Laparoscopists (AAGL). J Am Assoc Gynecol Laparosc 2007;7:9-15
- Kho RM, Hilger WS, Hentz JG et al. Robotic hysterectomy: technique and initial outcomes. Am J Obstet Gynecol 2007;197:113.e1-4
- Veljovich DS, Paley PJ, Drescher CW et al. Robotic surgery in gynecologic oncology: program initiation and outcomes after the first year with comparison with laparotomy for endometrial cancer staging. Am J Obstet Gynecol 2008;198:679.e1-9
- Magrina JF, Kho RM, Weaver AL et al. Robotic radical hysterectomy: comparison with laparoscopy and laparotomy. Gynecol Oncol 2008;109:86-91
- Boggess JF. Robotic-Assisted Hysterectomy for Endometrial Cancer. National Library of Medicine Archives. http://www.nlm.nih.gov/medlineplus/surgeryvideos.html Accessed September 22, 2009
- Advincula AP, Xu x, Goudeau S et al. Robotic-assisted laparoscopic myomectomy versus abdominal myomectomy: a comparison of short-term surgical outcomes and immediate costs. J Minim Invasive Gynecol 2007;14:698-705
- Advincula AP, Song A. The role of robotic surgery in gynecology. Curr Opin Obstet Gynecol 2007;19:331-336
- Geller EJ, Siddiqui NY, Wu JM et al. Short-term outcomes of robotic sacrocolpopexy compared with abdominal Sacrocolpopexy. Obstet Gynecol 2008;112:1201-1206
- Hemal AK, Kolla SB, Wadhwa P. Robotic reconstruction for recurrent supratrigonal vesicovaginal fistulas. J Urol 2008;180:981-985
- Leihan JP Jr, Kovanda C, Seshadri-Kreaden U. What is the learning curve for robotic assisted gynecologic surgery? J Minim Invasive Gynecol 2008;15:589-594
- Ota T, Degani A, Schwartzman D et al. A highly articulated robotic surgical system for minimally invasive surgery. Am Thorac Surg 2009;87:1253-1256
Published: 23 November 2009
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


